1 Wavelength R 1 N 2 1 N 2
enersection
Mar 18, 2026 · 7 min read
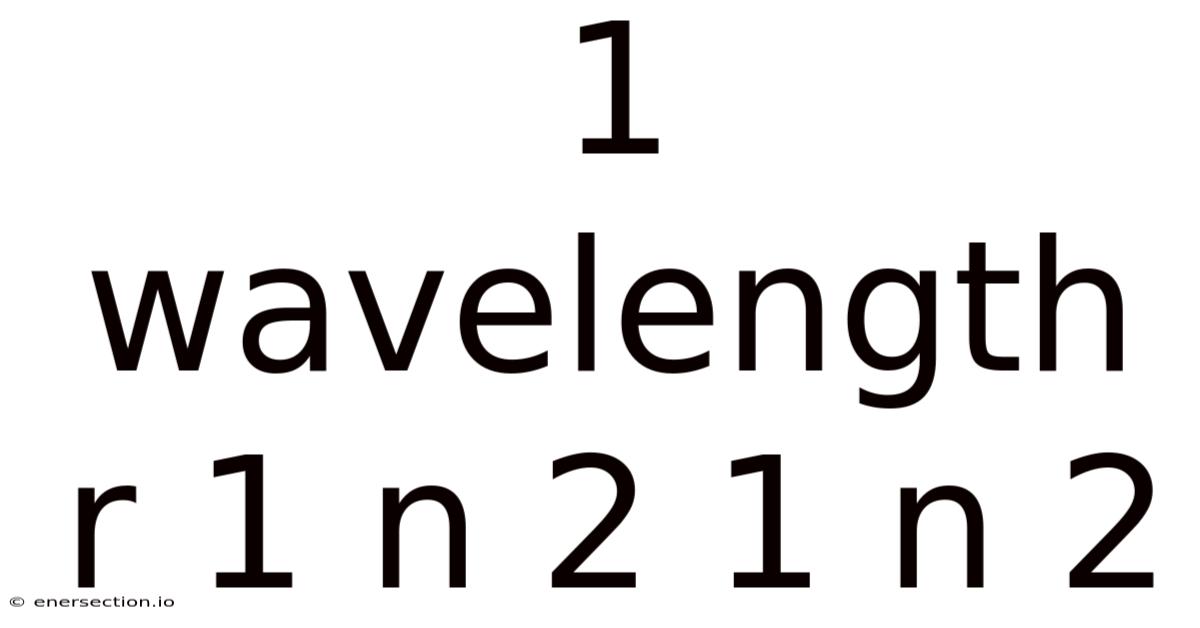
Table of Contents
Understanding the Wavelength Formula: 1/λ = R(1/n₁² - 1/n₂²)
The formula 1/λ = R(1/n₁² - 1/n₂²) represents one of the most fundamental relationships in atomic physics, describing the wavelengths of light emitted or absorbed when electrons transition between energy levels in hydrogen-like atoms. This equation, derived from the Bohr model of the atom, connects the visible world of light with the invisible quantum realm of atomic structure.
The Components of the Formula
To fully grasp this equation, we need to understand each component:
λ (lambda) represents the wavelength of the emitted or absorbed light, typically measured in nanometers (nm) or meters (m).
R is the Rydberg constant, approximately 1.097 × 10⁷ m⁻¹ for hydrogen. This universal constant relates to the fundamental properties of atomic structure.
n₁ and n₂ are integers representing the principal quantum numbers of the energy levels involved in the transition, where n₂ > n₁.
The formula can be rearranged to solve for wavelength: λ = 1/[R(1/n₁² - 1/n₂²)]
Physical Meaning and Applications
This formula describes the spectral lines produced when electrons jump between energy levels in atoms. When an electron moves from a higher energy level (n₂) to a lower one (n₁), it releases energy in the form of a photon with a specific wavelength determined by this equation.
The formula is particularly useful for:
- Calculating the wavelengths of spectral lines in hydrogen's emission spectrum
- Understanding the electronic structure of atoms
- Analyzing astronomical spectra to determine the composition of stars
- Developing technologies like lasers and spectroscopy instruments
Example Calculations
Let's calculate the wavelength of light emitted when an electron in a hydrogen atom transitions from n=3 to n=2:
1/λ = R(1/n₁² - 1/n₂²) 1/λ = 1.097 × 10⁷(1/2² - 1/3²) 1/λ = 1.097 × 10⁷(1/4 - 1/9) 1/λ = 1.097 × 10⁷(0.25 - 0.1111) 1/λ = 1.097 × 10⁷(0.1389) 1/λ = 1.523 × 10⁶ m⁻¹
Therefore: λ = 1/(1.523 × 10⁶) = 656.3 × 10⁻⁹ m = 656.3 nm
This wavelength corresponds to red light in the Balmer series, which is visible to the human eye.
The Rydberg Constant and Its Significance
The Rydberg constant R is derived from more fundamental constants:
R = (mₑe⁴)/(8ε₀²h³c)
where:
- mₑ is the electron mass
- e is the elementary charge
- ε₀ is the permittivity of free space
- h is Planck's constant
- c is the speed of light
This relationship shows how the formula connects microscopic quantum properties with macroscopic observable phenomena like light wavelengths.
Limitations and Extensions
While this formula works excellently for hydrogen and hydrogen-like ions (atoms with only one electron, such as He⁺ or Li²⁺), it has limitations:
- It doesn't account for electron-electron interactions in multi-electron atoms
- It doesn't include relativistic effects
- It doesn't consider fine structure splitting of spectral lines
For more complex atoms, quantum mechanical calculations using the Schrödinger equation provide more accurate results.
Historical Context
The formula was developed by Johannes Rydberg in 1888, building on earlier work by Johann Balmer. It predated the Bohr model of the atom and was initially an empirical relationship. The physical meaning became clear only after Niels Bohr proposed his atomic model in 1913, which explained why electrons occupy discrete energy levels.
Practical Applications in Modern Science
Today, this formula and its extensions are used in:
- Astronomy: Determining the composition of distant stars and galaxies by analyzing their light spectra
- Quantum computing: Understanding energy level transitions in quantum bits
- Chemical analysis: Identifying elements through their characteristic spectral lines
- Medical imaging: Technologies like MRI rely on principles of atomic energy transitions
Frequently Asked Questions
Q: Why must n₂ be greater than n₁ in the formula? A: The formula assumes emission of light (electron dropping to a lower energy level). If n₂ < n₁, it would represent absorption rather than emission, though the formula still works with appropriate sign changes.
Q: Can this formula be used for atoms other than hydrogen? A: Yes, but with modifications. For hydrogen-like ions (He⁺, Li²⁺, etc.), the formula becomes 1/λ = RZ²(1/n₁² - 1/n₂²), where Z is the atomic number.
Q: What are the visible spectral lines calculated using this formula? A: The Balmer series (transitions to n=2) produces visible light, including the famous red H-alpha line (n=3→2), blue-green H-beta line (n=4→2), and violet H-gamma line (n=5→2).
Conclusion
The formula 1/λ = R(1/n₁² - 1/n₂²) represents a beautiful bridge between the quantum world and our observable reality. It demonstrates how discrete atomic energy levels produce the continuous spectrum of light we see, from the colors in a rainbow to the light from distant stars. Understanding this relationship not only helps us calculate specific wavelengths but also provides insight into the fundamental nature of matter and energy. As quantum mechanics has evolved, this simple yet powerful formula remains a cornerstone of atomic physics, connecting theoretical predictions with experimental observations in laboratories and observatories around the world.
Expanding on the Formula’s Limitations and Refinements
While remarkably successful, the Rydberg formula possesses inherent limitations. As previously stated, it neglects relativistic effects, which become significant for heavier atoms where the electron’s velocity approaches a substantial fraction of the speed of light. These effects subtly alter the energy levels and, consequently, the calculated wavelengths. Similarly, the formula doesn’t account for fine structure splitting of spectral lines. This splitting arises from relativistic corrections and the interaction of the electron’s spin and orbital angular momentum with the magnetic field of the nucleus – a phenomenon described by the Zeeman effect. Ignoring these factors introduces inaccuracies, particularly when analyzing spectra of elements beyond hydrogen.
Furthermore, the formula’s empirical nature initially meant it was purely a descriptive tool. It didn’t inherently explain why electrons occupy specific energy levels. Bohr’s model provided this crucial explanatory framework, but more sophisticated quantum mechanical treatments, utilizing wave functions and solving the Schrödinger equation, offer far greater precision and a deeper understanding of atomic structure. These modern calculations incorporate electron-electron interactions and accurately predict spectral lines with significantly improved accuracy.
Beyond Hydrogen: Modifications and Extensions
The Rydberg formula’s adaptability extends beyond hydrogen-like ions. As noted, incorporating the atomic number (Z) allows it to predict spectral lines for ions like Helium (Z=2) and Lithium (Z=3). However, for more complex atoms with multiple electrons, the formula becomes increasingly cumbersome and less accurate. More advanced techniques, such as perturbation theory and Hartree-Fock calculations, are employed to determine energy levels and predict spectral transitions with greater fidelity. These methods often rely on sophisticated computational tools and databases of atomic data.
The Formula’s Legacy and Continued Relevance
Despite the advent of more complex quantum mechanical models, the Rydberg formula retains a vital place in scientific education and serves as a foundational concept. Its simplicity and elegance continue to illustrate the fundamental relationship between atomic structure and the electromagnetic spectrum. It’s a testament to the power of empirical observation and theoretical insight, demonstrating how a relatively simple equation can unlock profound understanding about the universe. The formula’s continued use in introductory physics courses underscores its enduring value as a gateway to more advanced concepts in atomic and quantum physics.
Conclusion
The Rydberg formula, born from meticulous observation and refined by theoretical advancements, stands as a landmark achievement in our understanding of the atom. While superseded by more comprehensive quantum mechanical models in its predictive power, it remains a cornerstone of atomic physics, offering a tangible connection between the microscopic world of electrons and the macroscopic world of light and color. Its legacy lies not just in its mathematical form, but in the conceptual leap it represented – the realization that the discrete nature of light is intimately linked to the quantized energy levels within atoms, forever changing our perception of matter and its interactions with the electromagnetic spectrum.
Latest Posts
Latest Posts
-
At What Temperature Does Salt Water Freeze
Mar 18, 2026
-
Does Coal Turn Into Diamonds Under Pressure
Mar 18, 2026
-
Slope And Y Intercept Word Problems
Mar 18, 2026
-
How To Make Ice Cubes Without An Ice Tray
Mar 18, 2026
-
Is Daisy Chaining Power Strips Bad
Mar 18, 2026
Related Post
Thank you for visiting our website which covers about 1 Wavelength R 1 N 2 1 N 2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.