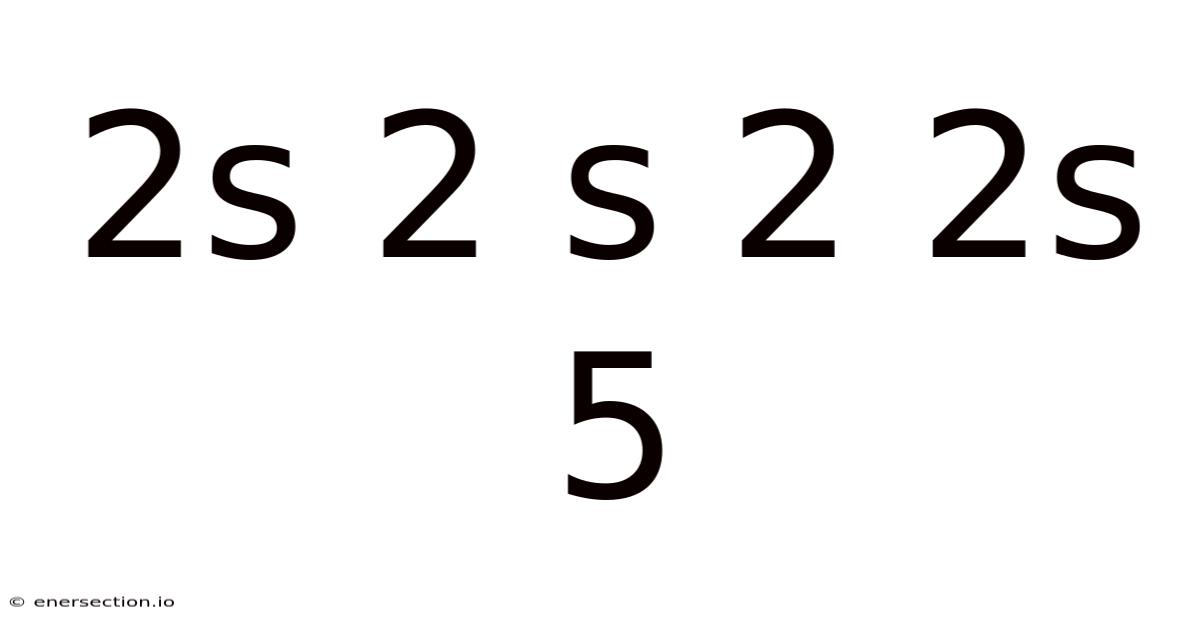The notation 2s 2 s 2 2s 5 appears cryptic at first glance, but it actually refers to a well-defined concept in atomic physics and quantum chemistry: the electron configuration of atoms. Understanding this notation is essential for anyone studying chemistry, physics, or materials science, as it reveals how electrons are distributed in atomic orbitals and directly influences an element's chemical behavior It's one of those things that adds up..
Understanding Electron Configuration
Electron configuration describes the arrangement of electrons in the orbitals of an atom. Each orbital is defined by a set of quantum numbers, and electrons fill these orbitals following specific rules: the Aufbau principle, Hund's rule, and the Pauli exclusion principle. The notation 2s 2 s 2 2s 5 can be interpreted as a shorthand for the distribution of electrons in various subshells That's the whole idea..
Breaking Down the Notation
Let's dissect the notation 2s 2 s 2 2s 5 step by step:
- 2s: This refers to the s-orbital in the second energy level (n=2). The s-orbital can hold a maximum of 2 electrons.
- s: This is likely a repetition or a reference to another s-orbital, possibly in a different context or energy level.
- 2: This could indicate the number of electrons in a particular orbital or subshell.
- 2s: Again, this points to the s-orbital in the second energy level.
- 5: This likely refers to the number of electrons in a specific orbital or the total number of electrons in a particular configuration.
Application in Atomic Structure
The electron configuration is crucial for understanding the periodic table and the chemical properties of elements. Take this: the configuration 2s 2 s 2 2s 5 might be used to describe the electron arrangement in certain ions or excited states of atoms. It helps predict how atoms will interact, bond, and participate in chemical reactions Most people skip this — try not to..
Importance in Chemistry and Physics
Electron configurations are foundational in explaining:
- The periodic trends in atomic size, ionization energy, and electronegativity.
- The formation of chemical bonds, such as ionic and covalent bonds.
- The magnetic properties of atoms and molecules.
- The spectral lines observed in atomic spectroscopy.
Frequently Asked Questions
Q: What does the notation 2s 2 s 2 2s 5 mean? A: This notation describes the distribution of electrons in atomic orbitals, specifically referencing the s-orbital in the second energy level and the number of electrons in various subshells Simple, but easy to overlook..
Q: How do I read electron configurations? A: Electron configurations are read by identifying the principal quantum number (n), the subshell letter (s, p, d, f), and the number of electrons in that subshell. As an example, 2s² means two electrons in the s-orbital of the second energy level And it works..
Q: Why is electron configuration important? A: It determines the chemical and physical properties of elements, influences bonding behavior, and explains periodic trends in the periodic table And it works..
Q: Can electron configurations change? A: Yes, electron configurations can change when atoms gain or lose electrons to form ions, or when electrons are excited to higher energy levels.
Conclusion
The notation 2s 2 s 2 2s 5 is a concise way to represent the complex arrangement of electrons in atomic orbitals. That's why by understanding this notation, students and researchers can gain deeper insights into atomic structure, chemical bonding, and the periodic behavior of elements. Mastery of electron configurations is essential for anyone pursuing studies in chemistry, physics, or related fields, as it forms the basis for predicting and explaining the behavior of matter at the atomic level But it adds up..
Beyond the Basics: Refining Electron Configuration Notation
While the presented notation offers a valuable starting point, it’s important to recognize that electron configurations can be represented with greater precision and detail. On top of that, the use of superscripts to denote the number of electrons within each orbital is standard practice. Now, the “5” in our example, for instance, could be interpreted in several ways. It might represent the total number of electrons in a specific shell, or it could be a placeholder for a more complex subshell designation – perhaps indicating a partially filled d or f orbital. As an example, “2s²” explicitly states that the 2s orbital contains two electrons.
More sophisticated notation utilizes boxes and arrows to visually depict electron filling patterns, a method known as Hund’s rules diagrams. Now, these diagrams clearly illustrate the principle of minimizing electron-electron repulsion, which dictates that electrons will individually occupy each orbital within a subshell before pairing up. This approach is particularly helpful when dealing with elements possessing multiple valence electrons That's the whole idea..
Adding to this, the concept of electron configuration extends beyond simple single-atom descriptions. Molecular electron configurations are crucial for understanding the structure and properties of molecules. These configurations account for the interactions between multiple atoms and the resulting bonding arrangements. Computational chemistry relies heavily on electron configuration principles to predict and simulate molecular behavior Practical, not theoretical..
This changes depending on context. Keep that in mind.
Expanding Applications and Modern Techniques
The principles of electron configuration aren’t confined to textbooks. That said, they are actively utilized in a wide range of modern scientific applications. Here's one way to look at it: in materials science, electron configurations are used to design new materials with specific electronic properties – crucial for developing semiconductors, superconductors, and advanced electronic devices. Here's the thing — similarly, in astrophysics, understanding the electron configurations of stars allows scientists to model their evolution and predict their behavior. Quantum chemistry employs sophisticated computational methods, such as Density Functional Theory (DFT), to accurately determine electron configurations and predict molecular properties with remarkable precision It's one of those things that adds up..
Not obvious, but once you see it — you'll see it everywhere.
Conclusion
Electron configuration, represented through various notations and visualized through diagrams, remains a cornerstone of modern chemistry and physics. While the initial example – “2s 2 s 2 2s 5” – provides a basic framework, a deeper understanding encompasses the nuances of orbital filling, the application of Hund’s rules, and the complexities of molecular configurations. Continual advancements in computational techniques and the expanding applications across diverse scientific fields check that the study of electron configuration will remain a vital tool for unraveling the fundamental nature of matter and its interactions.
This pedagogical challenge highlights a key tension: while the Aufbau principle provides a foundational scaffold, real-world electron configurations frequently deviate from the predicted order due to subtle energetic trade-offs between subshell stability and electron-electron repulsion. Elements like chromium (4s¹3d⁵) and copper (4s¹3d¹⁰) are classic exceptions where a half-filled or fully filled d-subshell offers greater stability, forcing an electron from the 4s to the 3d orbital. These exceptions are not mere curiosities but are critical for understanding the anomalous chemistry of transition metals, including their variable oxidation states and colorful coordination complexes Not complicated — just consistent..
Beyond isolated atoms and simple molecules, the concept evolves into band theory for extended solids. Here, the discrete energy levels of individual atoms blur into continuous energy bands—valence and conduction bands—whose structure, dictated by the collective electron configurations of all constituent atoms, determines a material’s metallic, semiconducting, or insulating character. This quantum-mechanical view is essential for engineering materials at the nanoscale, where quantum confinement alters band gaps and enables the design of quantum dots with tunable optical properties for displays and solar cells The details matter here. Less friction, more output..
On top of that, electron configuration serves as a bridge to deeper quantum phenomena. And the specific arrangement of electrons, particularly unpaired spins in partially filled orbitals, underpins magnetic properties—from the ferromagnetism in iron to the complex spin textures in quantum materials like topological insulators. In spectroscopy, the distinct energy differences between configurations correspond to specific wavelengths of absorbed or emitted light, allowing astronomers to deduce the elemental composition of distant stars and galaxies from their spectral fingerprints Not complicated — just consistent. Turns out it matters..
Conclusion
Thus, electron configuration transcends its origins as a simple bookkeeping device for atomic electrons. It is a dynamic framework that adapts to explain exceptions, scales up to describe collective behavior in solids, and connects to fundamental properties like magnetism and light interaction. Even so, from predicting the reactivity of a single element to designing the next generation of quantum materials and interpreting cosmic spectra, the principles of electron configuration remain an indispensable and evolving language. Its true power lies not in memorizing sequences, but in understanding the quantum mechanical forces that shape the electronic architecture of all matter, a perspective that continues to drive innovation across chemistry, physics, materials science, and astronomy Which is the point..