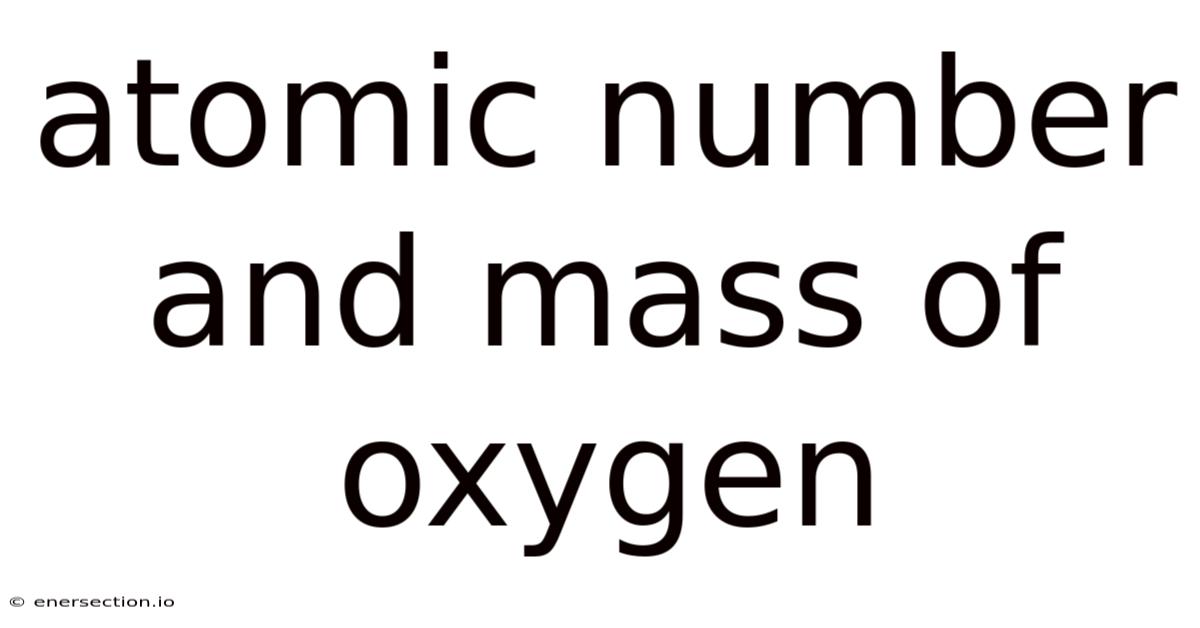The nuanced dance of atoms underpins the very fabric of our universe, yet few concepts resonate as profoundly as the atomic number and mass of oxygen. These fundamental parameters define the essence of an element, dictating its chemical behavior, physical properties, and role within the natural world. For oxygen, a cornerstone of life on Earth, understanding its atomic number and mass becomes a gateway to grasping its significance in biology, industry, and science as a whole. This article breaks down the nuances of these two attributes, exploring how they interplay to shape oxygen’s identity and its impact across diverse fields. By examining the interrelationships between atomic number, mass, and isotopic variations, we uncover a tapestry of information that reveals oxygen’s indispensable position in both natural and constructed environments.
Understanding Atomic Numbers and Mass in Chemistry
Atomic number serves as a cornerstone metric, representing the number of protons within an atom’s nucleus, which directly influences its chemical behavior. Oxygen, an element central to life, sits at the 8th position in the periodic table, reflecting its proton count of eight. Now, this number is not arbitrary; it arises from the fundamental principles of nuclear physics, where protons dictate the element’s identity. Here's the thing — the atomic number acts as a universal identifier, distinguishing oxygen from other elements and anchoring it within the periodic table’s framework. Similarly, mass number, though distinct from atomic number, complements this by accounting for neutrons, which contribute to an element’s total mass. While oxygen’s atomic mass is approximately 16 atomic mass units (amu), its precise composition includes two protons, six neutrons, and two electrons, resulting in a mass that balances stability and reactivity Which is the point..
The interplay between atomic number and mass number reveals oxygen’s unique position in the periodic table. As an oxygen atom, it balances between being relatively heavy yet chemically lightweight compared to heavier elements like carbon or nitrogen. This duality influences its reactivity, making it a versatile player in chemical reactions. Think about it: for instance, oxygen’s tendency to gain or lose electrons to achieve a stable electron configuration drives many biochemical processes, from cellular respiration to photosynthesis. Because of that, here, the atomic number’s role becomes important, as it dictates the element’s capacity to form bonds that sustain life. Understanding these concepts demands a grasp of how atomic structure translates into observable phenomena, bridging the abstract nature of particles with tangible outcomes.
The Significance of Oxygen’s Atomic Properties
Oxygen’s atomic properties extend beyond mere numbers, permeating every facet of its existence. The ability of oxygen molecules (O₂) to form stable bonds with carbon molecules underpins the formation of organic compounds, enabling the synthesis of proteins, DNA, and other biomolecules essential for biological systems. Still, its atomic number of eight positions it within the second period of the periodic table, where elements exhibit distinct chemical behaviors influenced by their nuclear configuration. This position places oxygen at the heart of its role as a respiratory gas, a key component in sustaining aerobic life forms. Beyond that, oxygen’s presence in the atmosphere, albeit trace, acts as a catalyst for combustion and weather patterns, shaping Earth’s climate systems.
The mass of oxygen, approximately 16 atomic mass units, contributes to its unique physical characteristics. This weight influences oxygen’s behavior in various contexts, from its use in fire extinguishers to its role in industrial processes like oxygenation in water treatment. Additionally, the mass contributes to oxygen’s ability to dissolve in water, a property critical for aquatic ecosystems. That said, this same mass also impacts oxygen’s solubility and reactivity, making it a subject of study in both biological and chemical sciences. The interplay between atomic number and mass thus becomes a lens through which oxygen’s multifaceted role is perceived, highlighting how fundamental constants govern its impact on the planet.
Not the most exciting part, but easily the most useful Small thing, real impact..
Isotopes and Variations in Oxygen’s Composition
While oxygen exists primarily as O₂, its isotopic composition introduces additional layers of complexity, further defining its properties and applications. Oxygen’s most common isotope, oxygen-16 (¹⁶O), constitutes about 99.8% of atmospheric oxygen, while oxygen-17 (¹⁷O) and oxygen-18 (¹⁸O) account for smaller percentages. Which means these isotopes arise from natural radioactive decay and environmental processes, resulting in slight variations in atomic mass and chemical behavior. Oxygen-16, with its even number of protons and neutrons, is the most stable isotope, making it prevalent in biological systems and industrial applications. Conversely, oxygen-17 and 18 exhibit unique properties that influence their utility in specific contexts, such as oxygen-18’s role in tracing human and animal migration patterns through isotopic analysis.
The study of these isotopes necessitates careful consideration of their implications, as even minor differences can alter an element’s behavior. Here's one way to look at it: oxygen-17’s presence in certain biological processes may affect metabolic rates, while oxygen-18’s isotopic signature helps scientists reconstruct past environmental conditions. Such nuances underscore the importance of precision in scientific inquiry, as even small variations can have cascading effects on ecosystems and human activities. By examining these isotopic variations, researchers gain deeper insights into oxygen’s role in both natural and engineered systems, reinforcing its status as a multifaceted element Most people skip this — try not to. Worth knowing..
Applications and Implications of Oxygen’s Properties
The practical applications of oxygen’s atomic number and mass extend far beyond theoretical understanding, influencing technology, medicine, and environmental conservation. In industry, oxygen is utilized as a reducing agent in manufacturing processes, while its role in combustion drives power generation and transportation. In medicine, oxygen therapy is critical for treating respiratory conditions, highlighting its biological significance. Environmental scientists use oxygen’s properties to monitor air quality, assess carbon dioxide levels, and study ocean acidification, which impacts marine life and climate regulation. Beyond that, oxygen’s presence in water systems plays a vital role in nutrient cycling, supporting aquatic food chains and biodiversity.
Beyond direct applications, oxygen’s characteristics also inform scientific research methodologies. Day to day, its stability and reactivity make it a benchmark for studying other elements, while its prevalence in natural atmospheres underscores its importance in atmospheric chemistry. Even so, in education, oxygen serves as a focal point for teaching fundamental chemistry concepts, bridging classroom learning with real-world relevance. The interplay between atomic number and mass thus permeates various disciplines, reinforcing oxygen’s centrality to scientific progress and practical implementation.
Conclusion: A Foundation for Understanding
In synthesizing the roles of atomic number and mass in oxygen’s profile, it becomes evident that these parameters are not mere numbers but keys unlocking deeper insights into the element’s essence. Oxygen’s atomic number
Oxygen’s atomic number, which defines its position in the periodic table, and its atomic mass, which determines its physical and chemical properties, together establish a framework that governs its interactions in diverse environments. Day to day, this intrinsic duality enables oxygen to function as both a reactive participant in chemical processes and a stable reference point for scientific measurement. To give you an idea, its atomic number of 8 ensures it forms two stable valence electrons, driving its role in bonding, while its isotopic variations (such as oxygen-16, 17, and 18) allow for precise tracking in fields ranging from climate science to biochemistry. This balance between simplicity and complexity underscores oxygen’s adaptability, making it indispensable in both natural systems and human innovation.
Not the most exciting part, but easily the most useful.
The implications of this adaptability extend to addressing contemporary challenges. In climate science, understanding oxygen’s isotopic signatures aids in modeling oceanic and atmospheric changes, while in healthcare, oxygen delivery systems are optimized through precise knowledge of its molecular behavior. Also worth noting, as humanity grapples with energy sustainability, oxygen’s role in combustion and fuel efficiency remains central to developing cleaner technologies. By bridging atomic-scale properties with macroscopic applications, oxygen exemplifies how fundamental scientific principles can translate into transformative solutions.
Pulling it all together, oxygen’s atomic number and mass are not just defining characteristics but dynamic forces that shape its identity and utility. This element, humble in its atomic structure yet vast in its influence, reminds us that even the most common elements hold the keys to understanding and advancing our world. Even so, as research continues to unravel new dimensions of oxygen’s behavior, from quantum-level interactions to large-scale environmental processes, its significance will only grow. That said, they reveal a deeper truth: that the properties of matter are rooted in quantifiable parameters, yet their manifestations are boundless. Oxygen, in all its isotopic and chemical forms, remains a cornerstone of scientific exploration—a testament to the power of precision, curiosity, and the relentless pursuit of knowledge Which is the point..