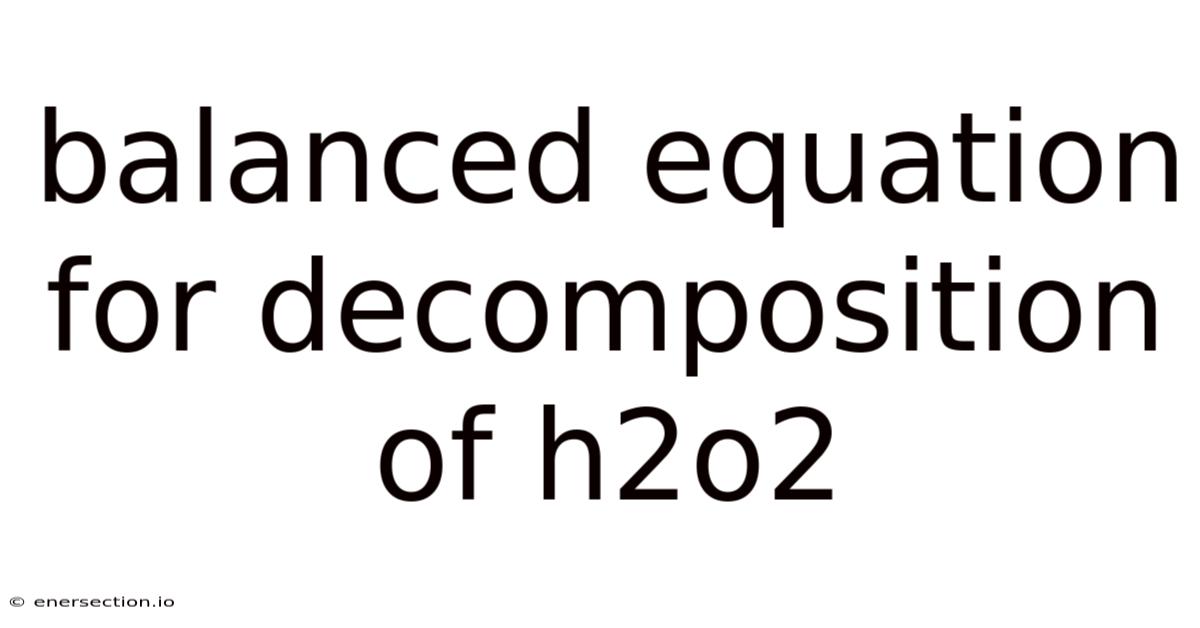The decomposition of hydrogen peroxide (H₂O₂) represents a critical process in various scientific, industrial, and environmental contexts. Understanding the precise mechanisms behind its decomposition is essential for grasping its implications in fields ranging from laboratory settings to natural ecosystems. In practice, this article walks through the intricacies of the balanced equation governing H₂O₂ decomposition, exploring its scientific foundations, practical applications, and the broader consequences of this reaction. The process begins with the recognition that H₂O₂, though seemingly stable under certain conditions, undergoes rapid transformation when exposed to specific triggers, such as heat, light, or catalytic agents. Which means whether in the synthesis of pharmaceuticals, the remediation of pollutants, or the production of hydrogen fuel cells, the precision required to align the equation’s stoichiometry with real-world conditions underscores its significance. What's more, the equation’s role extends beyond chemistry, influencing policy decisions regarding waste management and energy conservation. The balanced equation serves as a mathematical framework that quantifies these transformations, providing a reference point for chemists, engineers, and scientists aiming to harness H₂O₂ effectively. While often associated with its role as a powerful oxidizing agent, its chemical behavior is far more nuanced than its reputation suggests. By examining the interplay between molecular structure, thermodynamics, and environmental factors, readers will gain a comprehensive perspective on why this equation remains a cornerstone in chemistry education and application. Also, this decomposition is not merely a simple breakdown into water and oxygen; rather, it involves a complex interplay of oxidation and reduction reactions that dictate the efficiency and safety of the process. As the demand for sustainable solutions intensifies, the study of H₂O₂ decomposition becomes increasingly vital, highlighting the equation’s relevance as a tool for innovation and environmental stewardship. Through this lens, the balanced equation emerges not just as a formula but as a catalyst for understanding the principles that underpin chemical behavior, enabling advancements that shape both current practices and future technologies That's the part that actually makes a difference. That's the whole idea..
Not the most exciting part, but easily the most useful.
The balanced equation for the decomposition of hydrogen peroxide (H₂O₂) is 2H₂O₂ → 2H₂O + O₂, a reaction that encapsulates both the chemical transformation and the underlying principles that govern its occurrence. At its core, this equation illustrates the dual role of hydrogen peroxide as both an oxidizing agent and a substrate for decomposition, where the oxygen atoms within H₂O₂ are released as molecular oxygen, while hydrogen ions are transferred to water molecules. Now, the stoichiometry of the reaction—specifically the ratio of two moles of H₂O₂ yielding two moles of water and one mole of oxygen gas—reflects the conservation of mass and the stoichiometric precision required for the process to proceed efficiently. Still, the practical application of this equation often demands careful consideration of variables such as temperature, pressure, and the presence of catalysts, which can significantly influence the reaction rate and outcome. That's why for instance, elevated temperatures may accelerate the decomposition process, while the absence of certain catalysts might hinder it, leading to incomplete reactions or undesirable byproducts. In laboratory settings, this equation serves as a foundational reference, allowing researchers to predict the behavior of H₂O₂ under controlled conditions and optimize experimental outcomes. Beyond the laboratory, the equation finds utility in industrial applications where large-scale production or controlled environments are necessary. The synthesis of hydrogen peroxide itself relies heavily on this decomposition process, making it a critical link between chemical theory and industrial practice. That said, additionally, in environmental contexts, understanding the decomposition pathway helps in mitigating its potential hazards, such as the release of oxygen gas, which can pose risks if not managed properly. The equation thus acts as a bridge between theoretical knowledge and practical implementation, ensuring that the chemical principles are applied effectively. Its relevance extends beyond chemistry, influencing fields such as medicine, where H₂O₂ is both a therapeutic agent and a byproduct, and energy sectors, where efficient decomposition could contribute to cleaner energy solutions. Think about it: by mastering this equation, individuals and organizations gain the tools necessary to address challenges associated with H₂O₂, whether in the production of eco-friendly products or the management of hazardous waste. This equation’s versatility underscores its importance as a universal reference point, reinforcing its status as a cornerstone in both academic and applied disciplines Still holds up..
Subsequently, the decomposition of H₂O₂ is not confined to laboratory scenarios; it permeates everyday life through its dual role as both a reactive compound and a byproduct of various processes. In household environments, for example, accidental spills or improper handling of hydrogen peroxide can lead to unexpected outcomes, where the decomposition reaction might produce oxygen gas, which, if released uncontrolled, could pose safety concerns. Conversely, the controlled application of H₂O₂ in household cleaners or antiseptics relies on precise adherence to the balanced equation to ensure efficacy and safety
In the context of environmental systems, the decomposition of hydrogen peroxide plays a important role in natural and engineered processes. In wastewater treatment, for instance, H₂O₂ is often employed as an oxidizing agent to break down organic pollutants, with its decomposition governed by the same fundamental equation. Here, the reaction's efficiency depends on factors such as pH, temperature, and the presence of catalysts like iron or manganese ions, which can accelerate the breakdown process. Here's the thing — understanding these variables allows engineers to optimize treatment protocols, ensuring that the decomposition occurs at a rate that maximizes pollutant removal while minimizing the release of excess oxygen, which could disrupt aquatic ecosystems. Plus, similarly, in atmospheric chemistry, the decomposition of H₂O₂ contributes to the formation of hydroxyl radicals, which are crucial in the degradation of greenhouse gases and other atmospheric pollutants. This natural process underscores the equation's relevance in addressing global environmental challenges, such as climate change and air quality management. By applying the principles encapsulated in this equation, scientists and policymakers can develop strategies to harness or mitigate the effects of H₂O₂ decomposition in both natural and artificial systems, ensuring a balance between its benefits and potential risks Small thing, real impact..
The equation's applicability also extends to the field of materials science, where hydrogen peroxide is used in the synthesis of advanced materials, such as metal oxides and nanocomposites. In these processes, the controlled decomposition of H₂O₂ serves as a source of oxygen, facilitating the formation of desired compounds with specific properties. That's why this intersection of chemistry and materials science highlights the equation's role as a tool for innovation, driving advancements in technology and industry. Here, the equation provides a framework for predicting and controlling the reaction conditions, enabling researchers to tailor the properties of the materials to meet specific requirements. On the flip side, for example, in the production of titanium dioxide nanoparticles, the decomposition of H₂O₂ can influence the size, shape, and crystallinity of the resulting particles, which in turn affect their performance in applications like photocatalysis or solar cells. On top of that, the equation's principles are not limited to hydrogen peroxide alone; they serve as a model for understanding the decomposition of other peroxides and reactive oxygen species, further expanding its utility across diverse scientific domains.
At the end of the day, the decomposition of hydrogen peroxide, governed by its balanced chemical equation, is a phenomenon of profound significance that transcends disciplinary boundaries. From its foundational role in chemical education to its practical applications in industry, environmental management, and materials science, the equation serves as a unifying principle that connects theory with practice. Its versatility lies in its ability to adapt to varying conditions, whether in the controlled environment of a laboratory or the dynamic systems of the natural world. That's why by mastering this equation, individuals and organizations gain not only a deeper understanding of chemical processes but also the tools to address real-world challenges, from optimizing industrial production to mitigating environmental impacts. As science and technology continue to evolve, the principles encapsulated in this equation will remain a cornerstone, guiding innovation and ensuring the responsible application of chemical knowledge. In this way, the decomposition of H₂O₂ stands as a testament to the enduring relevance of fundamental chemical principles in shaping a sustainable and technologically advanced future.
Some disagree here. Fair enough.