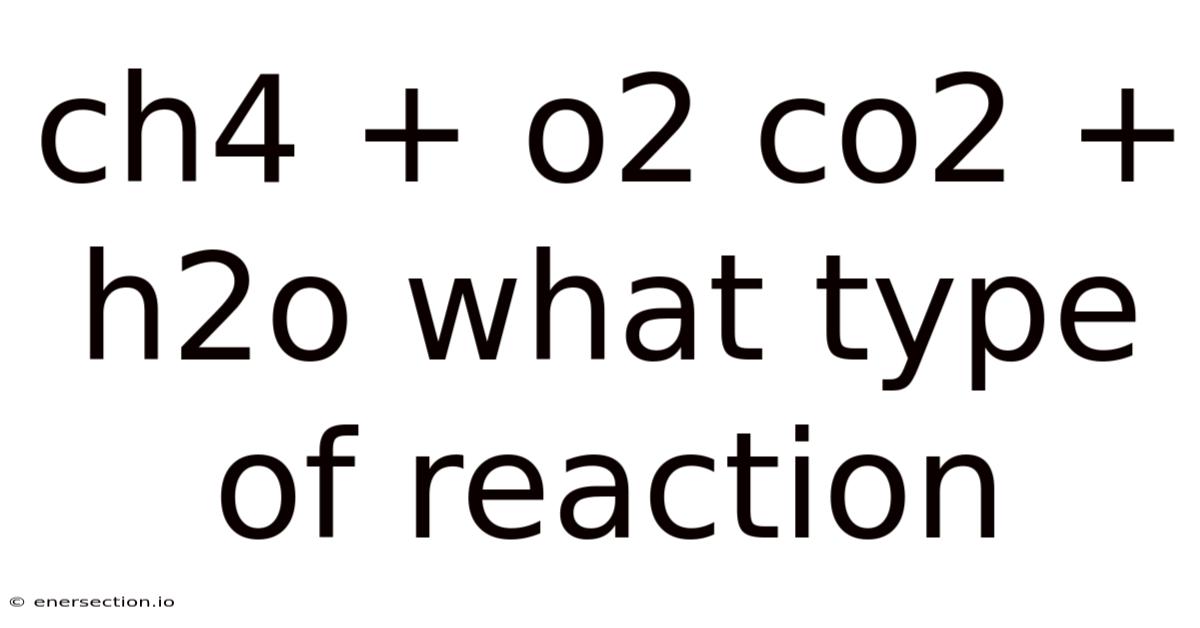The Chemistry of Combustion: Understanding CH4 + O2 → CO2 + H2O
When you strike a match to light a stove, ignite a campfire, or see the blue flame of a Bunsen burner in a lab, you are witnessing one of humanity’s oldest and most fundamental chemical processes: combustion. At its heart lies a simple yet profoundly important reaction, often written in its most iconic form as CH4 + O2 → CO2 + H2O. That's why this equation, representing the burning of methane, is more than just a string of chemical formulas; it is a cornerstone of energy production, a key player in Earth’s climate system, and a perfect example of a specific class of chemical reactions. This article will delve deep into the nature of this reaction, explaining not only what type it is but why it happens, the science behind it, and its vast implications for our world.
The Combustion Reaction Explained
The reaction CH4 + O2 → CO2 + H2O is the balanced chemical equation for the complete combustion of methane (CH4), the primary component of natural gas. Let’s break down the components:
- CH4 (Methane): A simple hydrocarbon, consisting of one carbon atom bonded to four hydrogen atoms. And it is a potent greenhouse gas and a major fuel source. * O2 (Oxygen): The oxidizing agent from the air we breathe. Combustion cannot occur without it. On the flip side, * CO2 (Carbon Dioxide): The product of carbon oxidation. * H2O (Water): The product of hydrogen oxidation, released here as vapor (steam).
The balanced equation requires stoichiometric coefficients: CH4 + 2O2 → CO2 + 2H2O. So this means one molecule of methane reacts with two molecules of oxygen gas to produce one molecule of carbon dioxide and two molecules of water. This precise ratio is critical for complete combustion.
What Type of Reaction Is It?
This reaction is classified as a combustion reaction, specifically the combustion of a hydrocarbon. Combustion is a high-temperature, exothermic (heat-releasing) redox reaction where a fuel (the reductant) reacts rapidly with an oxidant, typically oxygen, to produce oxidized products. In this case:
- Redox (Oxidation-Reduction): It is unequivocally a redox reaction.
- Oxidation: The carbon in methane (oxidation state -4) loses electrons as it bonds with more electronegative oxygen, increasing its oxidation state to +4 in CO2. The hydrogen (oxidation state +1) also loses electrons, but its oxidation state remains +1 in H2O; its "oxidation" is better described as being converted from a fuel-bound state to water.
- Reduction: The oxygen molecule (O2, oxidation state 0) gains electrons, with its oxidation state decreasing to -2 in both CO2 and H2O.
- Exothermic: The reaction releases a significant amount of energy—primarily as heat and light (the flame). Here's the thing — the energy stored in the strong covalent bonds of methane is greater than the energy stored in the bonds of the products (CO2 and H2O). The excess energy is liberated, making methane an excellent fuel.
- Synthesis? While it forms new compounds (CO2 and H2O) from simpler ones, it is not classified as a synthesis (or combination) reaction because it involves the breaking apart of the fuel and oxidant molecules and their recombination in a more complex, energy-releasing process. Synthesis reactions are typically A + B → AB.
- Decomposition? No, it is the opposite; simpler molecules combine to form more oxidized ones.
Most guides skip this. Don't Still holds up..
Because of this, the most precise classification is a combustion reaction, which is a subset of redox reactions No workaround needed..
The Scientific Principles at Play
The Mechanism of Complete Combustion
For combustion to be "complete," three elements—often called the "fire triangle"—must be present in the correct proportions:
- Fuel: Methane (CH4).
- Oxidizer: Oxygen (O2) from air (air is ~21% O2).
- Ignition Source: Sufficient heat/energy to overcome the activation energy barrier and initiate the chain reaction.
In a perfect scenario with adequate oxygen and mixing, the reaction proceeds through a radical chain mechanism. That said, the initial heat breaks some bonds, creating highly reactive free radicals (like •CH3, •H, •OH). These radicals collide with other molecules, propagating the reaction in a self-sustaining cascade until all fuel is converted to CO2 and H2O, releasing maximum energy and a typically blue, hot flame That's the part that actually makes a difference..
Incomplete Combustion: The Other Side of the Coin
When oxygen is limited or mixing is poor, incomplete combustion occurs. The reaction cannot proceed fully to CO2 and H2O. Instead, partially oxidized products form:
- Carbon Monoxide (CO): A highly toxic, colorless, odorless gas. Formed when carbon is only partially oxidized (oxidation state +2 instead of +4).
- Carbon (Soot, C): Unburned carbon particles that appear as black smoke or soot.
- Other Hydrocarbons (e.g., C2H2, C2H4): Unreacted or partially reacted fuel molecules.
The equation for incomplete combustion might look like: 2CH4 + 3O2 → 2CO + 4H2O (producing carbon monoxide) or CH4 + O2 → C + 2H2O (producing soot). Incomplete combustion is inefficient (wastes fuel), dangerous (produces CO), and polluting (produces soot).
The Environmental and Practical Significance
The Energy Engine
The exothermic nature of this reaction is why methane is a cornerstone of the global energy economy. The heat released from burning natural gas (mostly CH4) in power plants, home furnaces, and water heaters is harnessed to generate electricity, provide warmth, and cook food. The reaction’s efficiency and relatively lower carbon dioxide emission per unit of energy compared to coal or oil make it a "bridge fuel" in the transition to cleaner energy Simple, but easy to overlook..
The Climate Connection
The products of complete combustion are the two most significant greenhouse gases (GHGs) from human activity:
- Carbon Dioxide (CO2): The primary long-lived GHG driving anthropogenic climate change. The CO2 from burning fossil fuels like methane accumulates in the atmosphere, trapping heat and altering the planet’s energy balance.
- Water Vapor (H2O): The most abundant GHG, but its concentration in the atmosphere is primarily controlled by natural temperature cycles (via evaporation and
Water Vapor: A Secondary but Potent Player
While water vapor is indeed the most abundant greenhouse gas, its atmospheric concentration is largely a function of temperature rather than direct emissions. In large‑scale combustion facilities, the cumulative addition of water vapor can modestly shift the local radiative balance, especially when combined with the primary CO₂ signal. Nonetheless, the water vapor released in a furnace or power‑plant plume can locally raise humidity, which in turn can enhance the greenhouse effect in that region. This feedback loop is one reason why engineers strive for the highest possible combustion efficiency: the less fuel burned, the less water vapor (and CO₂) added to the atmosphere And that's really what it comes down to. Less friction, more output..
1. Pollutants Beyond CO₂ and H₂O
Even when a flame looks “clean,” a suite of trace pollutants may be forming:
| Pollutant | Formation Pathway | Environmental / Health Impact |
|---|---|---|
| Nitrogen Oxides (NOₓ) | High‑temperature oxidation of atmospheric N₂ (thermal NOₓ) or reaction of N₂ with O radicals (prompt NOₓ). Day to day, | Contribute to tropospheric ozone, smog, and acid rain; precursor to secondary particulate matter. |
| Unburned Hydrocarbons (UHCs) | Incomplete oxidation of CH₄ or larger alkanes, especially in low‑temperature zones. | React with NOₓ to form ozone; some are toxic or carcinogenic. Think about it: |
| Particulate Matter (PM₂. In real terms, ₅) | Soot nucleation from carbon‑rich zones; can also incorporate metals from fuel impurities. | Respiratory and cardiovascular disease; visibility reduction. |
| Formaldehyde (CH₂O) & Acetaldehyde (CH₃CHO) | Partial oxidation intermediates that escape quenching. | Irritants; formaldehyde is a known carcinogen. |
People argue about this. Here's where I land on it Worth keeping that in mind..
Modern combustion systems therefore incorporate after‑treatment technologies—selective catalytic reduction (SCR) for NOₓ, oxidation catalysts for UHCs, and electrostatic precipitators or fabric filters for PM—to meet stringent air‑quality regulations Easy to understand, harder to ignore..
2. Engineering Strategies for Near‑Perfect Combustion
a. Premixed vs. Diffusion Flames
- Premixed flames blend fuel and oxidizer before ignition, allowing a thin reaction zone and excellent temperature uniformity. This configuration minimizes hotspots that support NOₓ formation.
- Diffusion (or non‑premixed) flames rely on mixing during combustion, which can generate richer pockets and higher peak temperatures, raising NOₓ and soot yields.
Advanced burners (e.So g. , low‑NOₓ premixers) deliberately create a staged mixture that burns progressively, keeping peak temperatures below the threshold for significant thermal NOₓ production.
b. Flame‑Stabilization Techniques
- Swirl injectors impart angular momentum to the flow, creating a central recirculation zone that anchors the flame and promotes thorough mixing.
- Recirculating burners channel a fraction of exhaust gases back into the combustion chamber, diluting the fresh mixture, lowering flame temperature, and increasing overall efficiency.
c. Catalytic Combustion
Catalysts such as platinum or palladium alloys can lower the activation energy so that the reaction proceeds at temperatures well below the conventional flame temperature (≈ 1500 K). The result is a flameless or distributed combustion that virtually eliminates NOₓ and soot while retaining high thermal efficiency Easy to understand, harder to ignore. Worth knowing..
d. Real‑Time Sensing and Control
- O₂ sensors (lambda probes) monitor excess air and feed back to the fuel‑air ratio controller.
- Infrared gas analyzers track CO and unburned CH₄, prompting corrective actions before emissions exceed limits.
- Machine‑learning algorithms now predict optimal burner settings based on ambient temperature, pressure, and load, continuously nudging the system toward the stoichiometric sweet spot.
3. Methane Leakage: The “Upstream” Counterpart
Even before combustion, methane can escape from extraction, processing, and distribution infrastructure. Because CH₄ has a global warming potential (GWP) roughly 28‑34 times that of CO₂ over a 100‑year horizon, any leak undermines the climate advantage of burning natural gas instead of coal Easy to understand, harder to ignore..
Mitigation tactics include:
- Advanced leak detection using laser‑based open‑path spectroscopy or drone‑mounted sensors.
- Component upgrades (e.g., replacing aging seals, installing dry‑seal compressors).
- Regulatory frameworks that impose stringent leak‑rate thresholds and enforce rapid repair.
When the full life‑cycle emissions—upstream leaks plus downstream combustion—are accounted for, natural gas can still be cleaner than coal, but the margin shrinks dramatically if leak rates exceed ~1 % of total production.