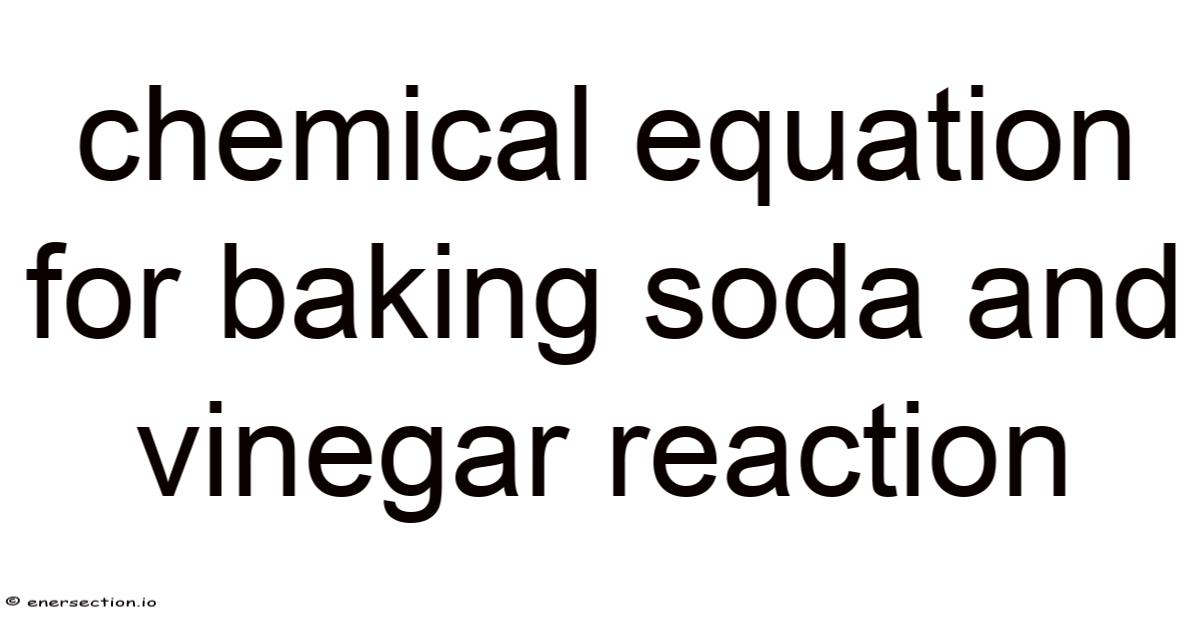Introduction
The classic baking soda and vinegar reaction is more than a fun classroom demonstration; it is a vivid example of an acid‑base neutralisation that produces carbon dioxide gas, water, and a soluble salt. Understanding the chemical equation behind this reaction helps students grasp fundamental concepts such as mole ratios, gas evolution, and the role of catalysts. This article breaks down the reaction step‑by‑step, explains the underlying chemistry, provides practical tips for safe experimentation, and answers common questions, all while keeping the content accessible to readers from any background And that's really what it comes down to..
The Core Chemical Equation
The overall balanced equation for the reaction between sodium bicarbonate (baking soda) and acetic acid (vinegar) is:
[ \boxed{\text{NaHCO}_3 (s) + \text{CH}_3\text{COOH} (aq) \rightarrow \text{NaCH}_3\text{COO} (aq) + \text{H}_2\text{O} (l) + \text{CO}_2 (g)} ]
- NaHCO₃ – sodium bicarbonate, a solid base.
- CH₃COOH – acetic acid, the main component of household vinegar (usually 5 % v/v).
- NaCH₃COO – sodium acetate, a soluble salt that remains dissolved in the liquid phase.
- H₂O – water, formed by the combination of a hydrogen ion from the acid and a hydroxide ion from the base.
- CO₂ – carbon dioxide gas, responsible for the characteristic fizz and bubbling.
The equation is already balanced: one mole of each reactant yields one mole of each product It's one of those things that adds up..
Step‑by‑Step Breakdown
1. Acid‑Base Neutralisation
When the bicarbonate ion (HCO₃⁻) encounters the acetate ion (CH₃COO⁻) from acetic acid, a proton transfer occurs:
[ \text{HCO}_3^- + \text{H}^+ \rightarrow \text{H}_2\text{O} + \text{CO}_2 \uparrow ]
The hydrogen ion (H⁺) comes from the dissociation of acetic acid in water:
[ \text{CH}_3\text{COOH} \rightleftharpoons \text{CH}_3\text{COO}^- + \text{H}^+ ]
2. Formation of Sodium Acetate
The sodium ion (Na⁺) from sodium bicarbonate pairs with the acetate ion (CH₃COO⁻) left over after proton donation, producing sodium acetate:
[ \text{Na}^+ + \text{CH}_3\text{COO}^- \rightarrow \text{NaCH}_3\text{COO} ]
3. Gas Evolution
The carbon dioxide generated is insoluble in water under normal conditions, so it escapes as bubbles. This rapid gas release creates the “volcano” effect often seen in experiments And it works..
Why the Reaction Is Exothermic
Although the overall enthalpy change is modest, the neutralisation of a weak acid (acetic acid) with a weak base (bicarbonate) releases a small amount of heat. Consider this: the temperature rise is usually only a few degrees Celsius, but it can be measured with a sensitive thermometer. This heat contributes to the vigorous bubbling and can be felt if the mixture is handled carefully.
Practical Laboratory Setup
Materials
| Item | Typical Quantity | Purpose |
|---|---|---|
| Sodium bicarbonate (baking soda) | 5 g | Reactant (solid) |
| White vinegar (5 % acetic acid) | 50 mL | Reactant (liquid) |
| Clear plastic bottle or graduated cylinder | 250 mL | Reaction vessel |
| Balloon or small plastic bag | 1 unit | Capture CO₂ (optional) |
| Safety goggles & gloves | – | Personal protection |
Procedure
- Measure 5 g of baking soda and place it into the bottle.
- Add 50 mL of vinegar quickly, then seal the opening with a balloon if you wish to collect the gas.
- Observe rapid bubbling and the balloon inflating.
- Record the time taken for the reaction to complete and note any temperature change.
Safety Considerations
- Eye protection is essential because the reaction can splash acidic liquid.
- Ventilation helps disperse CO₂, though the amounts produced are harmless in a typical classroom.
- Avoid ingestion of large quantities of either reactant; both are safe in small, household‑grade amounts but can cause irritation in excess.
Stoichiometric Calculations
Because the equation is 1:1, the amount of product formed is directly linked to the limiting reactant. Take this: if you start with 5 g of NaHCO₃:
- Molar mass of NaHCO₃ = 84 g mol⁻¹
- Moles of NaHCO₃ = 5 g ÷ 84 g mol⁻¹ ≈ 0.0595 mol
Acetic acid in 50 mL of 5 % vinegar:
- 5 % v/v ≈ 0.05 L × 1 g mL⁻¹ (density of vinegar ≈ 1 g mL⁻¹) = 0.05 g of pure acetic acid per mL → 0.05 g mL⁻¹ × 50 mL = 2.5 g
- Molar mass of CH₃COOH = 60 g mol⁻¹
- Moles of CH₃COOH = 2.5 g ÷ 60 g mol⁻¹ ≈ 0.0417 mol
Acetic acid is the limiting reactant. The theoretical yield of CO₂ is therefore 0.0417 mol, which corresponds to:
[ 0.0417 \text{ mol} \times 22.4 \text{ L mol}^{-1} \approx 0 Still holds up..
In practice, you will observe slightly less due to gas dissolution and incomplete mixing Small thing, real impact..
Scientific Explanation
Acid‑Base Theory
- Bronsted‑Lowry perspective: Acetic acid donates a proton (H⁺) to the bicarbonate ion, which acts as a base. The resulting conjugate base (acetate) pairs with the sodium cation, while the conjugate acid (carbonic acid, H₂CO₃) quickly decomposes into water and carbon dioxide.
[ \text{HCO}_3^- + \text{H}^+ \rightarrow \text{H}_2\text{CO}_3 \rightarrow \text{H}_2\text{O} + \text{CO}_2 \uparrow ]
- Le Chatelier’s principle: Removing CO₂ from the reaction mixture (by bubbling out) drives the equilibrium to the right, making the reaction appear “complete” even though the forward and reverse rates are still balanced at the microscopic level.
Gas Laws in Action
The rapid production of CO₂ allows a real‑world demonstration of Charles’s law (volume changes with temperature) and Boyle’s law (pressure changes with volume) when the gas is trapped in a balloon. As the balloon inflates, the internal pressure rises until it balances the atmospheric pressure plus the elastic tension of the balloon material.
Frequently Asked Questions
1. Why does the mixture fizz so violently?
The fizz is caused by CO₂ gas escaping the liquid phase. Because CO₂ is poorly soluble in water, it nucleates on any surface imperfections, forming bubbles that rise quickly And that's really what it comes down to. Less friction, more output..
2. Can I use other acids instead of vinegar?
Yes. Any strong or weak acid that provides H⁺ ions will react with sodium bicarbonate. Here's one way to look at it: lemon juice (citric acid) or dilute hydrochloric acid will produce the same overall products, though the rate and gas volume may differ.
3. What happens if I add too much baking soda?
Excess NaHCO₃ will remain unreacted, leaving a cloudy residue. The reaction will stop once all available H⁺ ions are consumed, so the amount of CO₂ produced is limited by the amount of acid Most people skip this — try not to..
4. Is the reaction reversible?
In a closed system where CO₂ is not allowed to escape, the reaction can reach an equilibrium that includes carbonic acid (H₂CO₃). That said, because CO₂ readily leaves the solution, the equilibrium is heavily shifted toward product formation Turns out it matters..
5. How can I capture the CO₂ for a science project?
Attach a balloon, a small plastic bag, or a graduated cylinder inverted over the reaction vessel. The gas will fill the container, allowing you to measure the volume produced Simple as that..
Applications Beyond the Classroom
- Cleaning: The effervescence helps dislodge grime in kitchen sinks and toilets.
- Fire extinguishers: Some dry‑chemical extinguishers use sodium bicarbonate to smother flames, although they do not rely on the vinegar reaction.
- DIY volcanoes: The dramatic eruption effect makes it a popular choice for educational demonstrations and themed parties.
- Carbonation experiments: By controlling the amount of CO₂ generated, students can explore the solubility of gases in water and the effect of temperature on carbonation.
Conclusion
The chemical equation for the baking soda and vinegar reaction encapsulates a simple yet powerful illustration of acid‑base neutralisation, gas evolution, and stoichiometry. That's why by balancing the equation, understanding each step, and applying safety‑first laboratory practices, learners can not only witness a spectacular fizz but also develop a deeper appreciation for the quantitative nature of chemistry. Whether you are a teacher planning a hands‑on activity, a student solving a chemistry problem, or a curious hobbyist experimenting at home, mastering this reaction provides a solid foundation for exploring more complex chemical processes Simple, but easy to overlook..