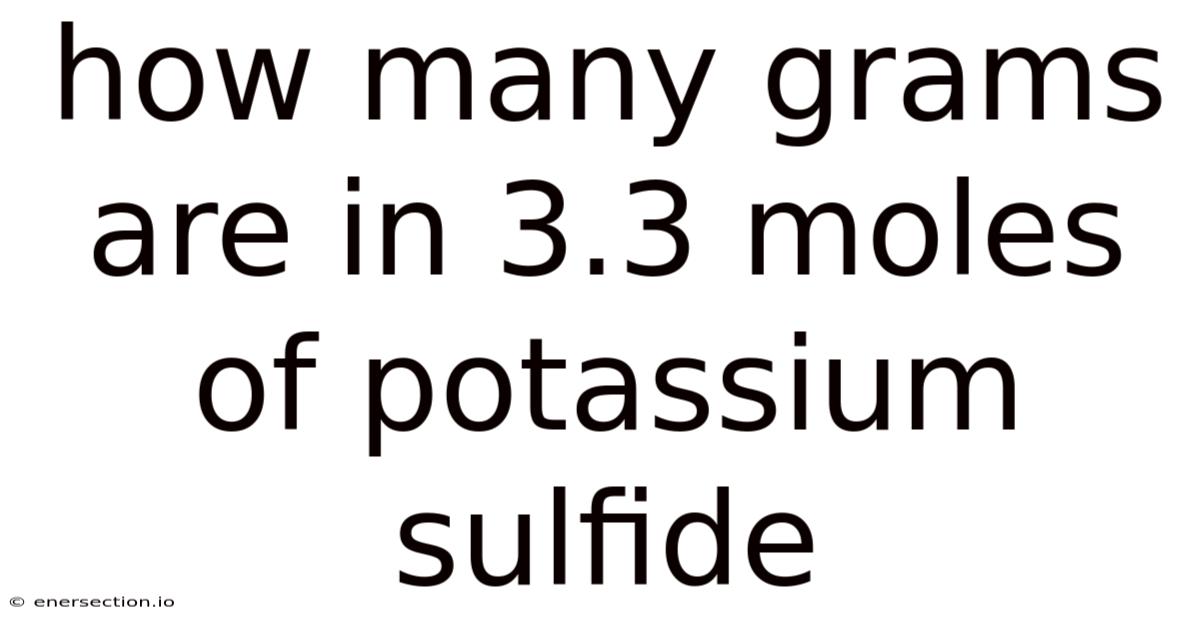The precise conversion between chemical quantities often serves as a cornerstone in scientific endeavors, where accuracy underpins everything from laboratory practices to industrial applications. Among these critical calculations lies one that demands meticulous attention to detail: determining how many grams correspond to a specified number of moles of a chemical compound. Such a task, while seemingly straightforward at first glance, involves navigating the detailed relationship between atomic weights, molar ratios, and fundamental unit conversions. In this context, understanding the precise relationship between moles and grams becomes critical, especially when dealing with elements like potassium sulfide, which has a real impact in various industrial processes and biological systems. Here's the thing — this article delves deeply into the mechanics behind this conversion, exploring the underlying principles that ensure reliability and precision. By breaking down the process step-by-step, we uncover not only the numerical outcome but also the underlying logic that makes such conversions both possible and trustworthy. The goal here is not merely to compute the answer but to illuminate the pathways that connect the abstract concept of moles to tangible measurements in the physical world The details matter here..
Molar Mass Calculation: The Foundation of Conversion
At the heart of converting moles to grams lies the concept of molar mass—a weighted average of the atomic weights of constituent elements. For potassium sulfide (K₂S), this involves summing the molar masses of potassium (K) and sulfur (S) across all atoms present in the formula. Potassium, an alkali metal, has a molar mass of approximately 39.10 g/mol, while sulfur, a non-metal, stands at 32.07 g/mol. That said, since potassium sulfide contains two potassium ions and one sulfur atom per formula unit, the calculation requires careful consideration of stoichiometry. The molar mass of K₂S is derived by multiplying the atomic mass of potassium by two (2 × 39.10) and adding the atomic mass of sulfur (32.07), resulting in a total of 78.23 g/mol. This foundational step is critical because even minor errors here can cascade into significant inaccuracies in the final result. This is genuinely important to verify atomic weights from reliable sources, as slight discrepancies might arise from rounding conventions or the choice of isotope used. Adding to this, understanding that molar mass represents the mass per mole allows chemists to bridge the gap between microscopic atomic structures and macroscopic measurements, making it indispensable for applications ranging from chemical synthesis to environmental monitoring. The process demands precision, as even a minor miscalculation can disrupt downstream calculations, emphasizing the need for thoroughness in handling such foundational data.
Application Context: Where Moles Find Their Role
Beyond theoretical understanding, the application of moles to grams becomes evident in numerous real-world scenarios where stoichiometry dictates outcomes. In laboratory settings, chemists often work with precise quantities, ensuring that molar ratios align perfectly with experimental designs. To give you an idea, synthesizing potassium sulfide requires exact stoichiometric proportions, where one mole of potassium sulfide produces a specific number of moles of potassium ions or sulfur atoms, each contributing to subsequent reactions or purification processes. Similarly, in industrial contexts, such as fertilizer production or mining operations, accurate mass conversions are vital for optimizing yields and minimizing waste. The concept extends beyond the laboratory into everyday life, influencing everything from cooking measurements to manufacturing processes that rely on precise chemical balances. Here, the conversion of moles to grams acts as a universal bridge, translating abstract numerical values into tangible quantities that can be measured and utilized effectively. Such applications underscore the practical significance of mastering this calculation, reinforcing its status as a fundamental skill across disciplines. The interplay between theory and practice necessitates a continuous reevaluation of how moles serve as a common language between different fields, fostering collaboration and consistency in scientific endeavors Worth keeping that in mind..
Common Misconceptions and Pitfalls to Avoid
Despite its importance, several misconceptions frequently hinder accurate application of molar conversions. One prevalent error involves miscalculating the molar mass itself, particularly when dealing with compounds containing multiple elements. Take this: conflating the atomic weights of potassium and sulfur or misapplying rounding rules can lead to off-target results. Another pitfall arises when neglecting the difference between molar mass and molecular weight, though in this context they are synonymous. Additionally, some may inadvertently confuse moles with grams directly, leading to confusion between the two units without proper context. There’s also a tendency to overlook the importance of significant figures when reporting results, resulting in either overly precise or understated numerical values that misrepresent the precision of the original data. Adding to this, certain individuals might mistakenly apply the same molar mass calculation to compounds with variable isotopic compositions, disregarding the need to specify the isotope used. Addressing these pitfalls requires vigilance, emphasizing the need for meticulous attention to detail and a thorough understanding of the foundational principles outlined earlier. Recognizing these potential pitfalls not only prevents errors but also enhances confidence in the process, ensuring that conversions remain reliable and consistent across diverse applications And that's really what it comes down to..
The Role of Precision in Scientific Integr
The Role of Precision in ScientificIntegrity
Precision in mole‑to‑gram conversions is not merely a matter of arithmetic; it underpins the credibility of experimental results and the reproducibility of scientific claims. When a chemist reports a yield of 0.842 mol of product, the corresponding mass must reflect the same level of certainty inherent in the measurement of moles. Over‑ or under‑estimating the molar mass by even a single significant figure can shift the calculated mass outside the experimental error margins, leading to false conclusions about reaction efficiency, purity, or stoichiometric balance.
One practical way to safeguard precision is to propagate uncertainties systematically. That said, 23 ± 0. Here's the thing — 008 ± 0. In real terms, , C = 12. In real terms, reporting the result as, for example, m = 10. g.Consider this: 001 g mol⁻¹), the combined standard uncertainty of the compound’s molar mass can be obtained via the root‑sum‑square method. Because of that, 001 g mol⁻¹, H = 1. Practically speaking, 011 ± 0. This uncertainty then combines with the uncertainty in the measured number of moles (often obtained from titration, gravimetry, or spectroscopic quantification) to yield a confidence interval for the final mass. If the molar mass is derived from atomic weights each accompanied by an uncertainty (e.05 g communicates both the value and its reliability, allowing peers to assess whether observed differences stem from genuine chemical variation or merely from rounding artifacts Worth knowing..
Another dimension of precision involves the choice of reference data. Also, using outdated values can introduce a systematic bias that accumulates over large‑scale calculations, such as those encountered in process engineering where ton‑scale batches are routine. Atomic weights are periodically revised by IUPAC to reflect improved isotopic abundance measurements. Laboratories should therefore maintain a version‑controlled database of constants and routinely verify that the molar masses employed in calculations correspond to the latest accepted standards.
Educational practices also play a role. Emphasizing the distinction between exact quantities (such as the stoichiometric coefficients in a balanced equation) and measured quantities (mass, volume, concentration) helps students internalize when to apply significant‑figure rules and when to retain extra digits for intermediate steps. Computational tools—spreadsheets, scripting languages, or dedicated chemical‑informatics software—can automate the tracking of significant figures and uncertainty propagation, reducing the likelihood of human error while reinforcing good habits.
Finally, precision fosters interdisciplinary communication. Similarly, environmental scientists estimating pollutant fluxes rely on accurate mole‑to‑gram conversions to model atmospheric chemistry or aquatic toxicity. In fields such as pharmacology, where a drug’s dosage is calculated from moles of active ingredient, a slight deviation in mass conversion can translate into under‑dosing or toxic overdosing. By upholding rigorous precision, scientists make sure the quantitative language of moles remains a reliable bridge linking theory, experimentation, and real‑world application.
Most guides skip this. Don't.
Conclusion
Mastering the conversion from moles to grams is far more than a routine calculation; it is a cornerstone of quantitative science that demands attention to molar‑mass accuracy, significant‑figure discipline, and uncertainty awareness. By recognizing common pitfalls, applying rigorous precision practices, and leveraging modern computational aids, researchers and practitioners can transform abstract mole quantities into trustworthy, measurable masses. This proficiency not only enhances the reliability of individual experiments but also strengthens the collaborative fabric of science, enabling consistent, reproducible advances across disciplines ranging from fundamental chemistry to industrial manufacturing and everyday life Nothing fancy..