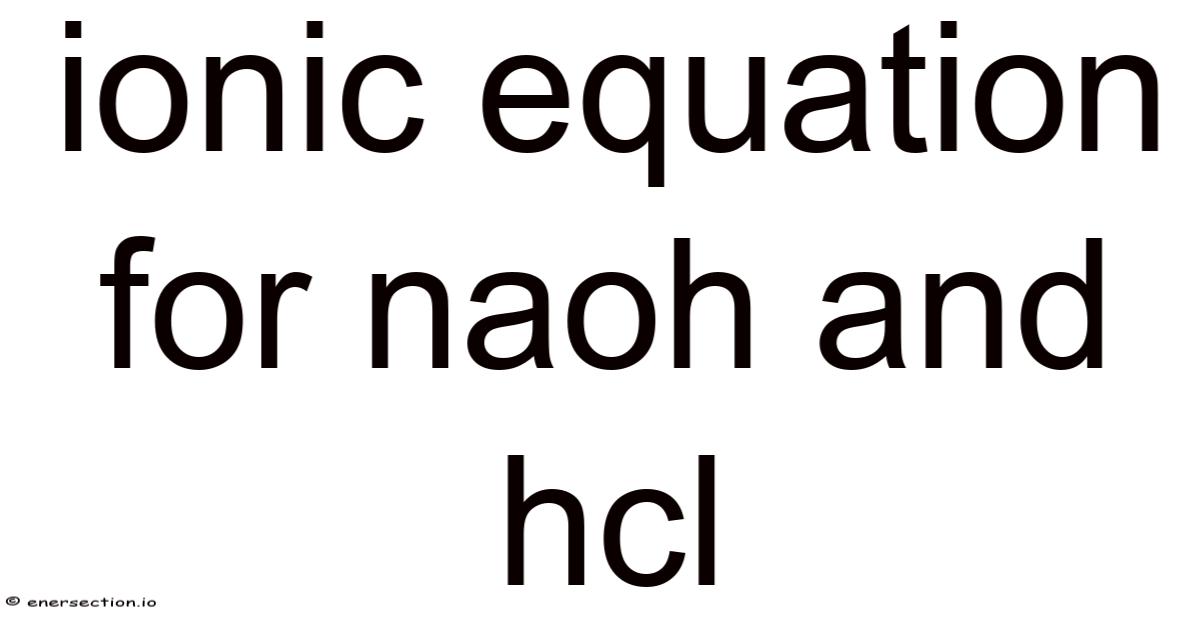The Ionic Equationfor NaOH and HCl: Understanding Neutralization at the Molecular Level
When sodium hydroxide (NaOH) reacts with hydrochloric acid (HCl), a classic example of a neutralization reaction occurs. This reaction is not only fundamental in chemistry but also widely applicable in real-world scenarios, from laboratory experiments to industrial processes. Practically speaking, by breaking down the reaction into its ionic components, we can better understand the role of each species and the principles governing acid-base neutralization. Consider this: the ionic equation for this reaction provides a clear representation of how the individual ions interact, offering deeper insight into the chemical process beyond the molecular level. This article will explore the ionic equation for NaOH and HCl, explain the steps to derive it, and highlight its significance in chemical education and practical applications Small thing, real impact..
Some disagree here. Fair enough.
Understanding the Molecular Equation
Before delving into the ionic equation, Start with the molecular equation — this one isn't optional. The reaction between sodium hydroxide and hydrochloric acid is represented as:
NaOH + HCl → NaCl + H₂O
This equation shows that one mole of sodium hydroxide reacts with one mole of hydrochloric acid to produce one mole of sodium chloride (table salt) and one mole of water. While this molecular equation is straightforward, it does not reveal the actual ionic interactions taking place in an aqueous solution. To fully grasp the chemistry behind this reaction, we need to examine the ionic equation.
The official docs gloss over this. That's a mistake.
Breaking Down the Ionic Equation
In an aqueous solution, both NaOH and HCl dissociate completely into their constituent ions. Sodium hydroxide, being a strong base, separates into sodium ions (Na⁺) and hydroxide ions (OH⁻). Similarly, hydrochloric acid, a strong acid, dissociates into hydrogen ions (H⁺) and chloride ions (Cl⁻). This dissociation is a critical step in forming the ionic equation.
Not obvious, but once you see it — you'll see it everywhere.
The total ionic equation for the reaction is written as:
Na⁺ + OH⁻ + H⁺ + Cl⁻ → Na⁺ + Cl⁻ + H₂O
Here, all the ions from the reactants and products are listed. On the flip side, not all ions participate in the actual chemical change. Sodium ions (Na⁺) and chloride ions (Cl⁻) remain unchanged throughout the reaction and are referred to as spectator ions. These ions do not contribute to the net chemical reaction and can be omitted to simplify the equation No workaround needed..
You'll probably want to bookmark this section Worth keeping that in mind..
Deriving the Net Ionic Equation
By removing the spectator ions (Na⁺ and Cl⁻) from the total ionic equation, we arrive at the net ionic equation. This equation focuses solely on the ions that undergo a chemical change:
OH⁻ + H⁺ → H₂O
This simplified equation clearly demonstrates the neutralization process. The hydroxide ion (OH⁻) from the base reacts with the hydrogen ion (H⁺) from the acid to form water (H₂O). This reaction is the core of acid-base neutralization, where an acid and a base combine to produce water and a salt Easy to understand, harder to ignore. No workaround needed..
Short version: it depends. Long version — keep reading.
Why the Ionic Equation Matters
The ionic equation is more than just a simplified version of the molecular equation. Now, it provides a precise understanding of the chemical process by highlighting the actual reacting species. In this case, it shows that the reaction is driven by the interaction between H⁺ and OH⁻ ions, which is a universal principle in acid-base chemistry. This concept is crucial for students and professionals alike, as it helps in predicting the outcomes of similar reactions involving other strong acids and bases.
The Role of Spectator Ions
Spectator ions
The Role of Spectator Ions
Spectator ions, while seemingly passive, play an important role in understanding the overall behavior of a solution. Their presence indicates the extent of dissociation of the reactants. Think about it: a solution containing a significant amount of spectator ions suggests that the reactants are strong electrolytes, meaning they readily dissociate into ions. Conversely, a solution with minimal spectator ions indicates weaker electrolytes, where only a small fraction of the reactant molecules dissociate. This information is vital in determining the concentration of ions in solution and predicting the electrical conductivity of the solution.
Applications in Chemical Analysis and Stoichiometry
The ability to write and understand ionic equations is invaluable in various chemical applications. Plus, in chemical analysis, ionic equations are used to identify and quantify ions present in a solution. In stoichiometry, the net ionic equation helps calculate the amounts of reactants and products involved in a reaction, considering only the species that participate in the chemical change. But for example, in titrations, understanding the ionic species involved allows for accurate determination of the concentration of acids and bases. This is particularly useful in quantitative analysis, where precise calculations are essential for determining the composition of a sample That's the part that actually makes a difference. That alone is useful..
Beyond Neutralization: Complex Reactions
The concept of ionic equations extends beyond simple neutralization reactions. By considering the ions involved, we can predict the formation of precipitates, the transfer of electrons, and the equilibrium constants of these reactions. They are equally important in understanding complex chemical processes, such as precipitation reactions, redox reactions, and acid-base reactions involving weak acids and bases. This understanding allows chemists to design and optimize chemical processes for various applications, from industrial manufacturing to environmental remediation.
Conclusion
The short version: while the molecular equation provides a convenient representation of chemical reactions, the ionic equation offers a more accurate and insightful understanding of the underlying chemical processes. Here's the thing — by breaking down the molecular equation into its constituent ions and identifying spectator ions, we can gain a deeper appreciation for the role of ions in chemical reactions. This knowledge is essential for a wide range of applications in chemistry, from basic laboratory techniques to complex industrial processes. Mastering the concept of ionic equations is a fundamental skill for any aspiring chemist, empowering them to analyze, predict, and manipulate chemical reactions with greater precision and understanding. The ability to discern the active species from the inert ones is a cornerstone of chemical comprehension, paving the way for more informed and effective chemical endeavors And that's really what it comes down to..
Building upon these insights, practical applications span diverse fields, demanding constant vigilance. Such knowledge empowers precise manipulation within complex systems Not complicated — just consistent..
Conclusion
Thus, grasping these principles remains crucial for advancing scientific progress and technological application. Mastery ensures effective engagement with the complex dynamics governing chemical interactions.
Continued Synthesis
This foundational understanding serves as a cornerstone, supporting further investigation and application in numerous contexts Which is the point..
Final Note
Understanding ion behavior thus concludes this discussion, emphasizing its universal relevance.
Stoichiometric calculations based on net ionic equations streamline experimental planning by focusing exclusively on the moles of species that undergo transformation. This precision reduces uncertainty in yield estimates and minimizes waste, particularly when reagents are expensive or environmental disposal must be limited. In titrimetry, for example, recognizing the essential ionic participants allows analysts to select indicators and conditions that sharpen endpoints, while in electrochemistry it clarifies how charge and mass relate through oxidation and reduction half-reactions.
These principles also scale elegantly from bench to plant. Practically speaking, process engineers use the same ionic logic to balance flows in continuous reactors, anticipate scaling or corrosion from sparingly soluble salts, and design ion-exchange or membrane separations that recover valuable components. Even in environmental chemistry, quantifying reactive ions guides the neutralization of acidic runoff, the precipitation of heavy metals, and the modeling of aquatic equilibria under changing temperature and pressure.
Conclusion
The bottom line: centering calculations on the ions that actively change reconciles theory with practice, converting abstract equations into reliable protocols. This disciplined focus sharpens prediction, conserves resources, and strengthens safety across laboratories and industries. By consistently distinguishing reactive participants from spectators, chemists equip themselves to innovate with clarity and responsibility, ensuring that each reaction is not only understood but also wisely applied The details matter here..