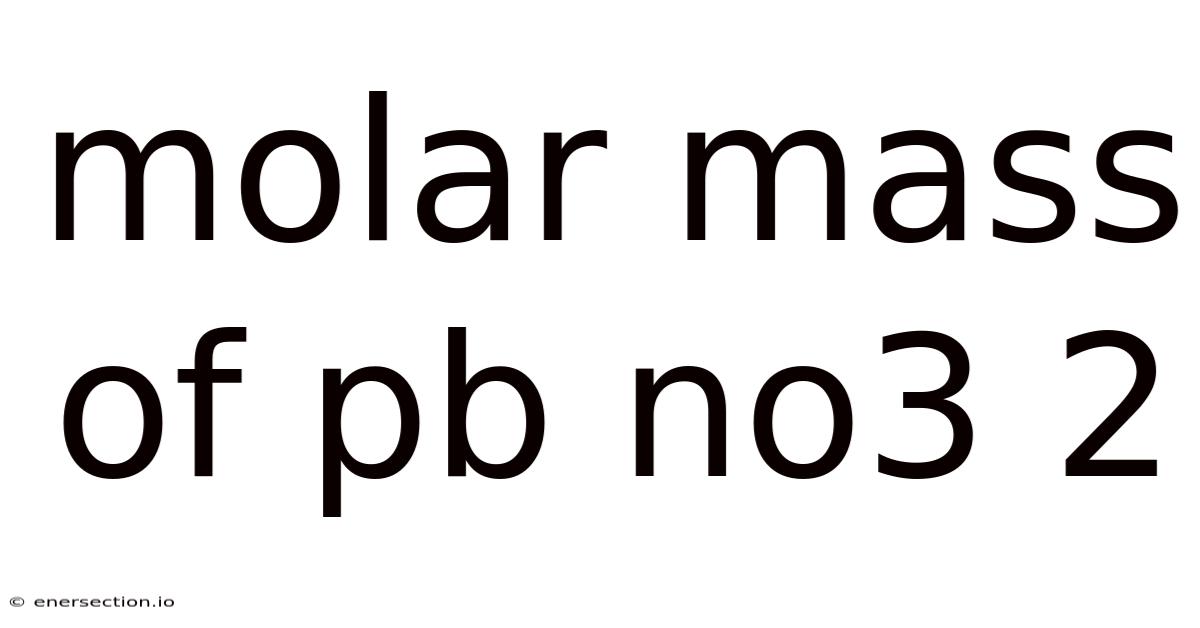Understanding the Molar Mass of Pb(NO₃)₂: A Step-by-Step Guide
The molar mass of Pb(NO₃)₂ is a fundamental concept in chemistry that plays a critical role in stoichiometry, chemical reactions, and laboratory calculations. On the flip side, lead(II) nitrate, with the chemical formula Pb(NO₃)₂, is a compound composed of lead ions (Pb²⁺) and nitrate ions (NO₃⁻). To determine its molar mass, we must sum the atomic masses of all the atoms present in the formula. Think about it: this process not only helps in quantifying substances but also ensures precision in experiments and industrial applications. In this article, we will explore how to calculate the molar mass of Pb(NO₃)₂, its significance, and common pitfalls to avoid The details matter here..
Breaking Down the Formula: Pb(NO₃)₂
Before diving into calculations, it’s essential to understand the structure of Pb(NO₃)₂. Here's the thing — the formula indicates one lead (Pb) atom and two nitrate (NO₃⁻) groups. Each nitrate group contains one nitrogen (N) atom and three oxygen (O) atoms.
- Lead (Pb): 1 atom
- Nitrogen (N): 2 atoms (1 from each nitrate group)
- Oxygen (O): 6 atoms (3 from each nitrate group)
This breakdown is crucial for accurate molar mass calculation.
Step-by-Step Calculation of Molar Mass
To calculate the molar mass of Pb(NO₃)₂, follow these steps:
-
Identify the atomic masses of each element from the periodic table:
- Lead (Pb): 207.2 g/mol
- Nitrogen (N): 14.01 g/mol
- Oxygen (O): 16.00 g/mol
-
Calculate the molar mass of one nitrate (NO₃⁻) group:
Nitrogen contributes 14.01 g/mol, and each oxygen contributes 16.00 g/mol.
Molar mass of NO₃⁻ = 14.01 + (3 × 16.00) = 62.01 g/mol -
Account for two nitrate groups in Pb(NO₃)₂:
Multiply the molar mass of one nitrate group by 2:
2 × 62.01 g/mol = 124.02 g/mol -
Add the molar mass of lead (Pb):
Total molar mass = Pb + 2 × NO₃⁻
= 207.2 + 124.02 = 331.22 g/mol
Thus, the molar mass of Pb(NO₃)₂ is 331.22 g/mol.
Why Is Molar Mass Important?
Understanding the molar mass of Pb(NO₃)₂ is vital for several reasons:
- Stoichiometric Calculations: It allows chemists to convert between grams and moles, essential for balancing chemical equations.
Plus, - Industrial Applications: Lead nitrate is used in pyrotechnics, glass production, and as a catalyst. Which means - Laboratory Work: Accurate molar mass ensures precise measurements in reactions, such as preparing solutions or synthesizing compounds. Knowing its molar mass aids in quality control and safety protocols.
No fluff here — just what actually works Less friction, more output..
Common Mistakes to Avoid
When calculating the molar mass of Pb(NO₃)₂, students often encounter these errors:
- Miscounting Atoms: Forgetting that the subscript outside the parentheses (the "2" in NO₃₂) applies to all atoms within the nitrate group.
In practice, - Incorrect Atomic Masses: Using outdated or rounded values for atomic weights instead of precise periodic table data. - Arithmetic Errors: Miscalculating sums or failing to multiply correctly when accounting for multiple atoms.
Always double-check your work by breaking down the formula and verifying each step But it adds up..
Scientific Explanation: Why Atomic Mass Matters
The atomic mass of an element represents the weighted average mass of its isotopes, measured in atomic mass units (u). 022 × 10²³). So when calculating molar mass, we convert these atomic masses to grams per mole (g/mol) using Avogadro’s number (6. For Pb(NO₃)₂, the molar mass reflects the combined mass of one lead atom, two nitrogen atoms, and six oxygen atoms. This value is critical because it bridges the microscopic world of atoms and molecules with macroscopic measurements used in laboratories That's the part that actually makes a difference..
Applications of Pb(NO₃)₂ Molar Mass
Knowing the molar mass of Pb(NO₃)₂ is not just an academic exercise. - Reaction Yields: In synthesis reactions, the molar mass helps determine the theoretical yield of products.
It has practical implications:
- Preparing Solutions: To create a solution of known concentration, you need to convert grams of Pb(NO₃)₂ to moles using its molar mass.
- Environmental Monitoring: Lead nitrate is regulated due to its toxicity; accurate measurements ensure compliance with safety standards.
Frequently Asked Questions (FAQ)
Q: What is the molar mass of Pb(NO₃)₂?
A: The molar mass is 331.22 g/mol, calculated by summing the atomic masses of Pb,
Q: What is the molar mass of Pb(NO₃)₂?
A: The molar mass is 331.22 g mol⁻¹, calculated by summing the atomic masses of Pb, N, and O in the formula.
Q: How do I use this value in a laboratory setting?
A: Convert the mass of lead nitrate you weigh into moles by dividing by 331.22 g mol⁻¹. Then, use the mole ratio from the balanced equation to determine how much of another reactant or product is needed or will be produced Which is the point..
Q: Can lead nitrate be safely handled in a standard chemistry lab?
A: Lead nitrate is toxic and a potential carcinogen. Always use it in a fume hood, wear appropriate personal protective equipment, and follow institutional safety guidelines The details matter here..
Q: What are common industrial uses of lead nitrate?
A: Lead nitrate is employed in pyrotechnics for bright green flames, in the production of glass and ceramics, as a catalyst in certain organic reactions, and historically in the manufacture of leaded gasoline (now largely phased out).
Q: Are there alternative sources of lead for industrial processes?
A: Yes, lead salts such as lead(II) acetate or lead(II) sulfate are also used, but each has distinct solubility, reactivity, and cost profiles that influence selection.
Conclusion
Calculating the molar mass of a compound like Pb(NO₃)₂ may seem like a routine exercise, yet it is a cornerstone of chemical practice. A precise molar mass links the abstract world of atomic theory to tangible laboratory work: it allows chemists to measure substances accurately, predict reaction outcomes, and maintain safety standards in both research and industry. Still, by understanding the step‑by‑step breakdown of the formula, avoiding common pitfalls, and appreciating the broader context—ranging from pyrotechnics to environmental regulations—students and professionals alike gain a deeper respect for the meticulous nature of stoichiometry. Remember that every mole you calculate is a bridge between the microscopic and macroscopic realms, and mastering this bridge equips you with a powerful tool for scientific inquiry and practical application The details matter here. Less friction, more output..
Understanding the relationship between molar mass and theoretical yield is essential for scientists aiming to optimize chemical processes efficiently. This concept not only guides laboratory experiments but also informs industrial applications, where precision can impact both performance and safety. By integrating these principles, researchers confirm that each reaction proceeds as intended, minimizing waste and maximizing results And that's really what it comes down to. That alone is useful..
Environmental considerations further underscore the importance of accurate measurements. Regulations surrounding substances like lead nitrate highlight the need for vigilance in handling and using materials that could pose health or ecological risks. Staying informed about such guidelines helps maintain a safe and compliant laboratory environment.
Worth adding, exploring broader applications reveals how foundational knowledge in molar mass underpins innovations across fields. Whether in manufacturing, education, or research, this principle remains a vital thread connecting theory and practice.
Simply put, mastering the interplay of molar mass and yield calculations empowers chemists to figure out complex challenges with confidence. This continuous learning reinforces the value of precision in science and its real‑world implications.