Net Ionic Equation For Hydrolysis Of Na2co3
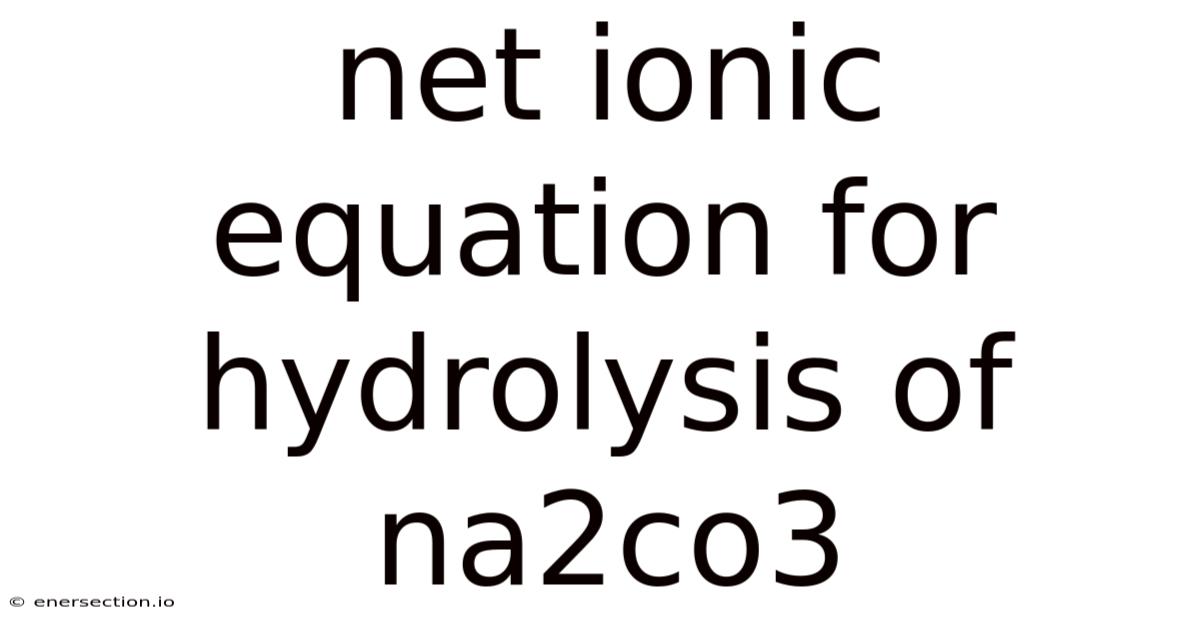
Netionic equation for hydrolysis of Na₂CO₃ – This article explains step‑by‑step how to write the net ionic equation for the hydrolysis of sodium carbonate, breaks down the underlying chemistry, and answers common questions that students and educators often encounter.
Introduction
Sodium carbonate (Na₂CO₃) is a widely used alkaline salt in laboratories, industry, and everyday life. When dissolved in water it undergoes hydrolysis, producing carbonate ions that react with water to generate bicarbonate and hydroxide ions. Understanding the net ionic equation for hydrolysis of Na₂CO₃ is essential for predicting solution pH, performing titration calculations, and designing buffer systems. This guide walks you through the logical process, from writing the full molecular equation to isolating the species that actually participate in the reaction.
The Reaction in Context
What Happens When Na₂CO₃ Dissolves?
- Dissociation into ions – Na₂CO₃(s) → 2 Na⁺(aq) + CO₃²⁻(aq)
- Hydrolysis of the carbonate ion – CO₃²⁻(aq) + H₂O(l) ⇌ HCO₃⁻(aq) + OH⁻(aq)
The hydroxide ions released are responsible for the basic nature of the solution. The sodium ions, being spectators, do not affect the acid‑base equilibrium and therefore do not appear in the net ionic equation.
Steps to Derive the Net Ionic Equation
1. Write the Complete Molecular Equation
Start with the reaction between solid sodium carbonate and water: Na₂CO₃(s) + H₂O(l) → 2 Na⁺(aq) + CO₃²⁻(aq) + H₂O(l)
Since water is both a reactant and a product in the dissolution process, it is usually omitted from the ionic representation.
2. Convert to Full Ionic Equation
Break all strong electrolytes into their constituent ions:
Na₂CO₃(s) → 2 Na⁺(aq) + CO₃²⁻(aq)
The hydrolysis step is already ionic: CO₃²⁻(aq) + H₂O(l) ⇌ HCO₃⁻(aq) + OH⁻(aq)
Combine them:
2 Na⁺(aq) + CO₃²⁻(aq) + H₂O(l) ⇌ 2 Na⁺(aq) + HCO₃⁻(aq) + OH⁻(aq)
3. Cancel Spectator Ions
Spectator ions are those that appear unchanged on both sides of the equation. Here, Na⁺ appears twice on the left and twice on the right, so they cancel out.
Resulting net ionic equation:
CO₃²⁻(aq) + H₂O(l) ⇌ HCO₃⁻(aq) + OH⁻(aq)
This is the net ionic equation for hydrolysis of Na₂CO₃. ### 4. Emphasize the Directionality
Because the reaction is reversible, the double arrow (⇌) indicates that both forward and reverse processes occur. In practice, the forward direction dominates under typical aqueous conditions, producing a basic solution.
Scientific Explanation ### Acid‑Base Perspective
The carbonate ion (CO₃²⁻) acts as a base according to the Brønsted‑Lowry definition, accepting a proton from water:
CO₃²⁻ + H₂O → HCO₃⁻ + OH⁻
The produced hydroxide ions increase the pH, making sodium carbonate solutions alkaline (pH ≈ 11.6 for a 0.1 M solution).
Equilibrium Constant
The equilibrium constant (Kₕ) for this hydrolysis can be derived from the Kₐ values of carbonic acid. Since CO₃²⁻ is the conjugate base of HCO₃⁻, its basicity is related to the second dissociation constant (Kₐ₂ ≈ 4.8 × 10⁻¹¹). The corresponding K_b for CO₃²⁻ is:
K_b = K_w / Kₐ₂ ≈ 1.0 × 10⁻¹⁴ / 4.8 × 10⁻¹¹ ≈ 2.1 × 10⁻⁴
A modest K_b confirms that the reaction proceeds appreciably but not completely, which is why the equilibrium arrow is used.
Role of Ionic Strength
In concentrated solutions, the activity coefficients of ions decrease, slightly altering the observed pH. However, the net ionic equation remains valid because it focuses on the stoichiometric species rather than concentration effects.
Frequently Asked Questions (FAQ)
Q1: Why are sodium ions excluded from the net ionic equation?
A: Sodium ions are spectator ions; they do not participate in the chemical change that defines the hydrolysis reaction. Removing them simplifies the equation to the essential acid‑base process.
Q2: Can the net ionic equation be written with a single arrow instead of a double arrow?
A: While many textbooks use a single forward arrow to emphasize the predominant direction, the hydrolysis of carbonate is reversible. Using a double arrow (⇌) accurately reflects the equilibrium nature of the process.
Q3: How does the hydrolysis of Na₂CO₃ compare to that of NaHCO₃? A: Sodium bicarbonate (NaHCO₃) hydrolyzes according to:
HCO₃⁻ + H₂O ⇌ H₂CO₃ + OH⁻
Both reactions generate OH⁻, but the equilibrium constants differ. Na₂CO₃ produces a more strongly basic solution because CO₃²⁻ is a stronger base than HCO₃⁻.
Q4: Does the presence of other ions affect the hydrolysis?
A: Yes. High ionic strength can shield charges and alter activity coefficients, slightly shifting the equilibrium position. However
Understanding the directionality of carbonate hydrolysis deepens our grasp of solution chemistry and its implications in both laboratory settings and industrial applications. This phenomenon not only clarifies how equilibrium works in complex systems but also highlights the subtle interplay between acid-base behavior and ion interactions. By recognizing the driving forces behind these reactions, chemists can better predict outcomes in titrations, buffer design, and environmental pH management.
In summary, the directionality in these equilibria underscores the importance of considering both stoichiometry and thermodynamic stability. The careful balance of ions and the influence of constants like Kₐ and K_b guide precise interpretations of what occurs at the molecular level. This knowledge empowers scientists to manipulate reactions efficiently, whether in analytical chemistry or sustainable resource management.
Concluding this exploration, appreciating the nuances of reaction arrows and equilibrium constants enriches our ability to navigate the intricate dance of molecules in aqueous environments. Such insights remain vital for advancing both theoretical understanding and practical applications in chemistry.
Building on this discussion, it’s clear that the nuances of writing net ionic equations extend beyond mere notation—they reflect a deeper comprehension of reaction mechanisms and equilibrium dynamics. Mastering these concepts allows researchers to anticipate shifts in solution behavior, especially when comparing different salts like Na₂CO₃ and NaHCO₃. Each case presents unique tendencies in acidity, hydrolysis, and ionic interactions, reinforcing the value of precise stoichiometric analysis.
Moreover, the role of constants such as Kₐ and K_b becomes increasingly pivotal as experiments progress toward real-world applications. These values help chemists not only explain observed phenomena but also predict how systems will respond under varying conditions. This predictive power is essential for fields ranging from environmental science to pharmaceuticals, where reaction control is critical.
In essence, the study of these equilibria underscores the interconnectedness of theory and practice. Each detail, from the exclusion of non‑involved ions to the influence of ionic strength, shapes our ability to manipulate and interpret chemical behavior. Recognizing these subtleties empowers scientists to design more effective experiments and solutions to complex challenges.
In conclusion, delving into these topics deepens our appreciation for the elegance of chemical systems and equips us with the tools necessary for precise scientific inquiry. The journey through stoichiometry and equilibrium is both intellectually rewarding and practically indispensable.
The ability to dissect and predict ionic behavior through net ionic equations is not merely an academic exercise; it is a cornerstone of modern chemical innovation. As industries increasingly prioritize sustainability and precision, the principles discussed—such as the manipulation of acid-base equilibria or the design of buffer systems—become indispensable tools. For instance, in water treatment, understanding how carbonate or bicarbonate ions interact with other species allows for the efficient removal of contaminants or the stabilization of pH in sensitive ecosystems. Similarly, in pharmaceutical development, precise control over ionic interactions can enhance drug solubility, bioavailability, and stability, directly impacting therapeutic efficacy.
The interplay between theoretical knowledge and applied science is further exemplified in emerging fields like green chemistry. Here, the precise management of ionic species can reduce waste, lower energy consumption, and minimize the environmental footprint of chemical processes. By leveraging the predictive power of equilibrium constants, researchers can design reactions that proceed with minimal byproducts, aligning with the principles of atom economy and green synthesis. This underscores how a deep understanding of net ionic behavior transcends laboratory settings, offering scalable solutions to global challenges.
Ultimately, the study of these equilibria is a testament to the power of chemical literacy. It bridges the gap between abstract molecular interactions and tangible outcomes, enabling scientists to engineer solutions that are both efficient and environmentally responsible. As new technologies and methodologies continue to evolve, the foundational principles of stoichiometry and equilibrium will remain vital, guiding advancements in chemistry that shape the future of science and society. The journey through these concepts is not just about mastering equations or constants—it is about cultivating a mindset that values curiosity, precision, and the relentless pursuit of understanding in an ever-changing world.
Latest Posts
Latest Posts
-
Does Ice Expand When It Freezes
Mar 26, 2026
-
How To Do Integrals On Ti 84 Plus
Mar 26, 2026
-
Rewrite The Following Expression In Terms Of The Given Function
Mar 26, 2026
-
How To Do Factorials On Ti 84
Mar 26, 2026
-
How To Put Games On The Ti 84 Plus
Mar 26, 2026