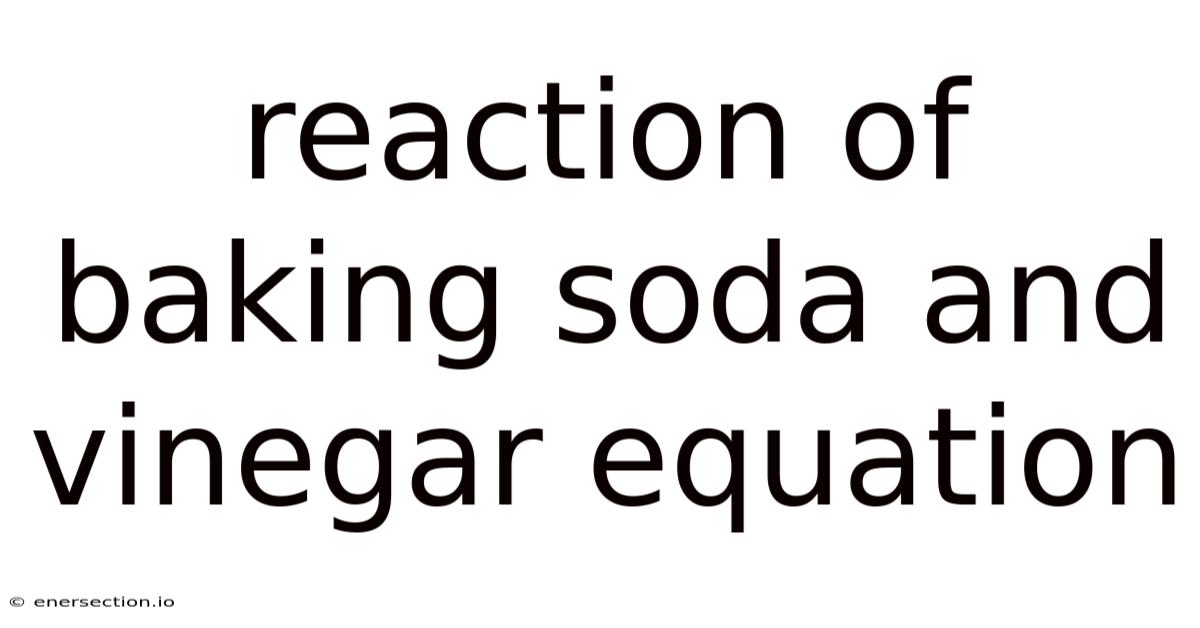Understanding the reaction of baking soda and vinegar is a fascinating journey into the world of chemistry, especially for students, educators, and curious minds eager to explore how everyday substances interact. In real terms, this simple yet powerful reaction not only demonstrates the principles of acid-base chemistry but also serves as a foundational concept in science education. In this article, we will delve deep into the science behind this classic experiment, its practical applications, and the significance it holds in both classroom learning and real-life scenarios Worth knowing..
The reaction of baking soda and vinegar is one of the most accessible demonstrations of chemical change in the world. Consider this: when these two common household items are combined, they trigger a rapid and visible transformation. This reaction is not only a fun activity for students but also a valuable teaching tool that helps demystify complex chemical concepts. Let’s explore what happens when baking soda, which is a base, meets vinegar, which is an acid, and how this interaction leads to a bubbly, exciting process.
At the heart of this reaction lies the interaction between sodium bicarbonate, the primary component of baking soda, and acetic acid, the main constituent of vinegar. When these two substances come into contact, they initiate a series of chemical transformations that result in the production of carbon dioxide gas, water, and sodium acetate. This process is not only safe but also highly educational, making it an ideal choice for science lessons across various educational levels.
To understand the reaction more clearly, it’s important to recognize the chemical formulas involved. Baking soda is composed of sodium bicarbonate, which can be represented as NaHCO₃, while vinegar contains acetic acid, with the formula CH₃COOH. When these two react, the following equation takes place:
Not obvious, but once you see it — you'll see it everywhere.
NaHCO₃ + CH₃COOH → CO₂ + H₂O + NaCH₃COO
This equation simplifies to the formation of carbon dioxide gas, water, and a sodium acetate salt. The presence of carbon dioxide is what causes the bubbles you see during the reaction, making it a visually engaging demonstration of chemical change Still holds up..
But why does this reaction occur? According to Le Chatelier’s Principle, when an acid and a base are mixed, the system shifts to counteract the change, leading to the formation of new substances. Baking soda is a base, and vinegar is an acid. The answer lies in the nature of acids and bases. In this case, the reaction is exothermic, meaning it releases heat, which is why the mixture often feels warm to the touch And that's really what it comes down to. Simple as that..
Easier said than done, but still worth knowing.
For students learning about acid-base reactions, this experiment is an excellent opportunity to observe the effects of neutralization. The reaction between baking soda and vinegar is a classic example of an acid-base neutralization reaction, which is a fundamental concept in chemistry. It helps students grasp the idea that certain substances can react to form new compounds while releasing energy in the process.
The reaction does not stop at just the immediate chemical change. That's why it also highlights the importance of understanding safety protocols when conducting such experiments. Although the reaction is generally safe, Use appropriate protective gear and ensure proper ventilation — this one isn't optional. This emphasizes the need for responsible scientific practice, especially in educational settings.
Another key aspect of this reaction is the role of gas production. Because of that, the carbon dioxide gas generated during the reaction is what creates the bubbling effect observed in the mixture. In practice, this gas is released because the acid in vinegar reacts with the base in baking soda, forming water and the salt sodium acetate. The continuous bubbling can be observed for several minutes, depending on the amount of reactants used and the rate of mixing No workaround needed..
Not obvious, but once you see it — you'll see it everywhere.
For those interested in expanding their understanding, it’s worth noting that this reaction is just one of many examples of acid-base reactions. Now, by exploring variations—such as using different concentrations of baking soda or vinegar—students can observe how changes in the amount of reactants affect the rate and intensity of the reaction. This hands-on approach reinforces the concept of scientific experimentation and the importance of controlled variables Worth keeping that in mind..
Beyond the classroom, the reaction between baking soda and vinegar has practical applications in everyday life. To give you an idea, it can be used in cleaning products, where the carbon dioxide produced helps lift dirt and grime. It also plays a role in food preservation and household cleaning, where the reaction can be harnessed to create effective and safe solutions Not complicated — just consistent..
In the realm of environmental science, understanding this reaction also helps explain natural processes. And for example, similar acid-base reactions occur in the human body, such as in the digestion of food. Also, the stomach, which contains a weak acid, reacts with the alkali in food to break down nutrients efficiently. This analogy not only strengthens students’ comprehension of chemistry but also connects it to real-world biological functions.
The scientific significance of this reaction extends to its role in chemical education. Teachers often use this experiment to introduce students to the concept of chemical equations and the laws of conservation of mass. By breaking down the reaction step by step, educators can help students visualize how atoms rearrange during a chemical change. This visual and interactive method enhances memory retention and fosters a deeper understanding of scientific principles.
Beyond that, the reaction is a great example of observational learning. Students who participate in this experiment develop critical thinking skills as they predict what will happen, observe the changes, and analyze the results. This process encourages curiosity and a scientific mindset, which are essential for future learning in STEM fields.
It is also important to highlight the cultural and historical context of this reaction. While the combination of baking soda and vinegar is a modern discovery, the principles behind it have been understood for centuries. Still, ancient civilizations recognized the power of acids and bases, using them in various applications from medicine to construction. By exploring this reaction, students can appreciate the evolution of scientific knowledge and its impact on society.
In addition to its educational value, this reaction has inspired countless experiments and variations. Take this: adding food coloring to the mixture can help visualize the reaction more clearly, while using different types of vinegar or baking soda can reveal how different substances interact. These variations not only make the learning experience more engaging but also encourage creativity and experimentation.
The emotional connection this experiment fosters is another key point. There’s something truly rewarding about watching a reaction unfold before your eyes. The sight of bubbles forming and the mixture transforming into a fizzy solution can spark excitement and a sense of accomplishment. This emotional engagement is crucial in keeping students motivated and interested in science.
As we explore the reaction of baking soda and vinegar, it becomes clear that this simple experiment is far more than just a fun activity. On top of that, it is a powerful tool for teaching scientific concepts, promoting hands-on learning, and fostering a deeper appreciation for chemistry. Whether you are a student, a teacher, or simply a curious learner, understanding this reaction can enhance your knowledge and inspire further exploration.
At the end of the day, the reaction of baking soda and vinegar is a testament to the beauty of chemistry in everyday life. By embracing this experiment, we not only gain a better understanding of the material but also cultivate a lifelong interest in science. It illustrates the principles of acid-base reactions, provides a safe and engaging way to learn, and offers a foundation for more advanced scientific studies. Let this article be the starting point for your journey into the fascinating world of chemistry.