The Half Life Of Plutonium 239 Is 24300 Years
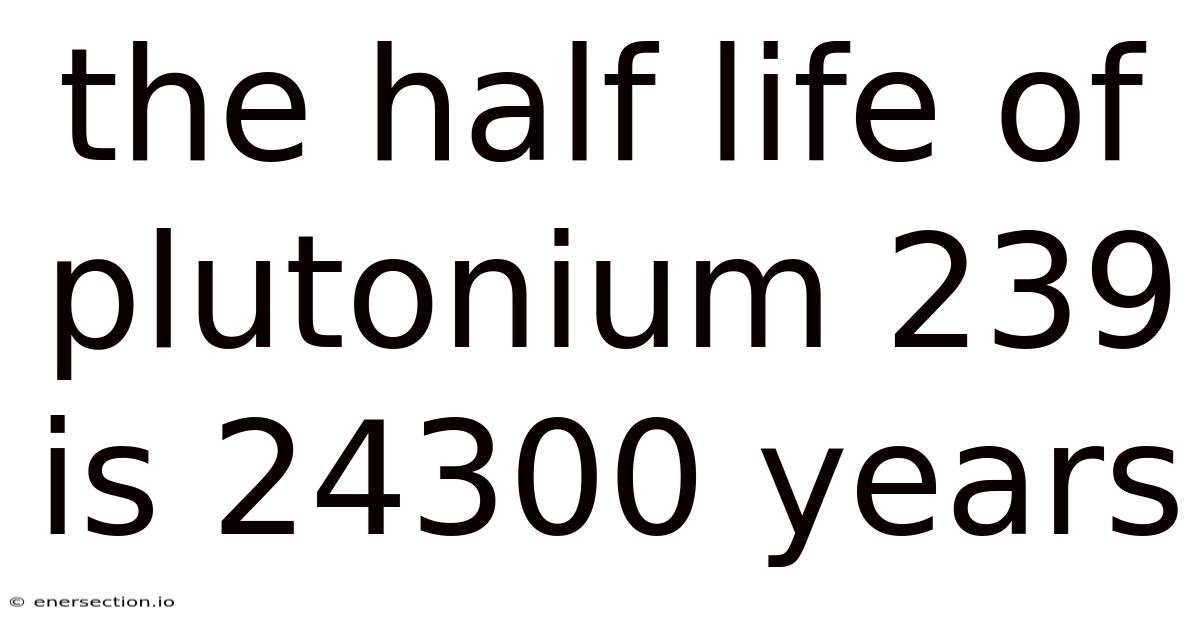
Plutonium-239 is one of the most significant isotopes in the world of nuclear science and energy production. With a half-life of 24,300 years, it plays a crucial role in both the creation of nuclear weapons and the generation of nuclear power. Understanding its properties, behavior, and implications is essential for anyone interested in nuclear physics, environmental science, or global security.
What is Plutonium-239?
Plutonium-239 is a radioactive isotope of plutonium, a heavy, silvery-gray metal. It is primarily produced in nuclear reactors when uranium-238 captures a neutron and undergoes a series of nuclear reactions. This isotope is fissile, meaning it can sustain a nuclear chain reaction, making it highly valuable for both energy production and weapons development.
The Significance of Its Half-Life
The half-life of a radioactive substance is the time it takes for half of its atoms to decay into other elements. For plutonium-239, this period is 24,300 years. This long half-life means that it remains hazardous for an extremely long time, posing challenges for storage, disposal, and environmental safety. Unlike shorter-lived isotopes, which decay quickly and lose their radioactivity, plutonium-239 remains a persistent threat, requiring secure containment for millennia.
How Does Plutonium-239 Decay?
Plutonium-239 undergoes alpha decay, a process in which it emits an alpha particle (two protons and two neutrons) and transforms into uranium-235. This decay process releases energy and ionizing radiation, which can be harmful to living organisms. The slow decay rate of plutonium-239 means that its radiation levels decrease gradually, but it remains dangerous for thousands of years.
Applications of Plutonium-239
Despite its risks, plutonium-239 has several important applications. In nuclear reactors, it is used as fuel to generate electricity. In nuclear weapons, it serves as a key component due to its ability to sustain a chain reaction. Additionally, plutonium-239 is used in some space missions as a power source for long-duration missions where solar energy is insufficient.
Environmental and Health Concerns
The long half-life of plutonium-239 raises significant environmental and health concerns. If released into the environment, it can contaminate soil and water, posing risks to ecosystems and human health. Inhalation or ingestion of plutonium-239 can lead to serious health issues, including cancer, due to its radioactive properties. This is why strict regulations govern its handling, storage, and disposal.
Safe Storage and Disposal
Given its long half-life, safe storage of plutonium-239 is a major challenge. It must be kept in secure facilities designed to prevent leaks or theft. Deep geological repositories, where radioactive waste is buried deep underground, are considered one of the safest options for long-term storage. However, the effectiveness of these methods over thousands of years remains a topic of ongoing research and debate.
Global Implications
The existence of plutonium-239 has significant global implications. Its use in nuclear weapons has shaped international relations and security policies. The Treaty on the Non-Proliferation of Nuclear Weapons (NPT) aims to prevent the spread of nuclear weapons and promote peaceful uses of nuclear energy. However, the dual-use nature of plutonium-239—its potential for both energy production and weapons development—continues to be a source of tension and concern.
Frequently Asked Questions
Why is the half-life of plutonium-239 so important?
The half-life determines how long the isotope remains radioactive and hazardous. With a half-life of 24,300 years, plutonium-239 requires long-term management and poses challenges for disposal.
Can plutonium-239 be destroyed or neutralized?
Plutonium-239 cannot be destroyed, but it can be transmuted into other isotopes through nuclear reactions. However, this process is complex and not yet widely implemented.
How is plutonium-239 different from other isotopes of plutonium?
Plutonium-239 is fissile, meaning it can sustain a nuclear chain reaction, unlike other isotopes such as plutonium-240, which is less suitable for weapons or reactor fuel.
What happens if plutonium-239 is released into the environment?
It can contaminate soil and water, posing long-term risks to ecosystems and human health. Its mobility in the environment depends on factors like chemical form and local conditions.
Is plutonium-239 naturally occurring?
Plutonium-239 is primarily man-made, produced in nuclear reactors. Trace amounts can be found in nature due to cosmic ray interactions, but these are negligible compared to human-produced quantities.
Conclusion
Plutonium-239, with its 24,300-year half-life, is a powerful yet challenging element. Its applications in energy and defense are significant, but so are the risks it poses to the environment and human health. Understanding its properties and managing its use responsibly is crucial for harnessing its benefits while minimizing its dangers. As technology and policy evolve, the global community must continue to address the challenges posed by this long-lived isotope.
Latest Posts
Latest Posts
-
Is Dissolving A Chemical Change Or Physical Change
Mar 20, 2026
-
Chi Square Test Of Homogeneity Vs Independence
Mar 20, 2026
-
How Are Power Work And Energy Related
Mar 20, 2026
-
How To Calculate Power From Voltage And Current
Mar 20, 2026
-
Range And Domain Of A Parabola
Mar 20, 2026