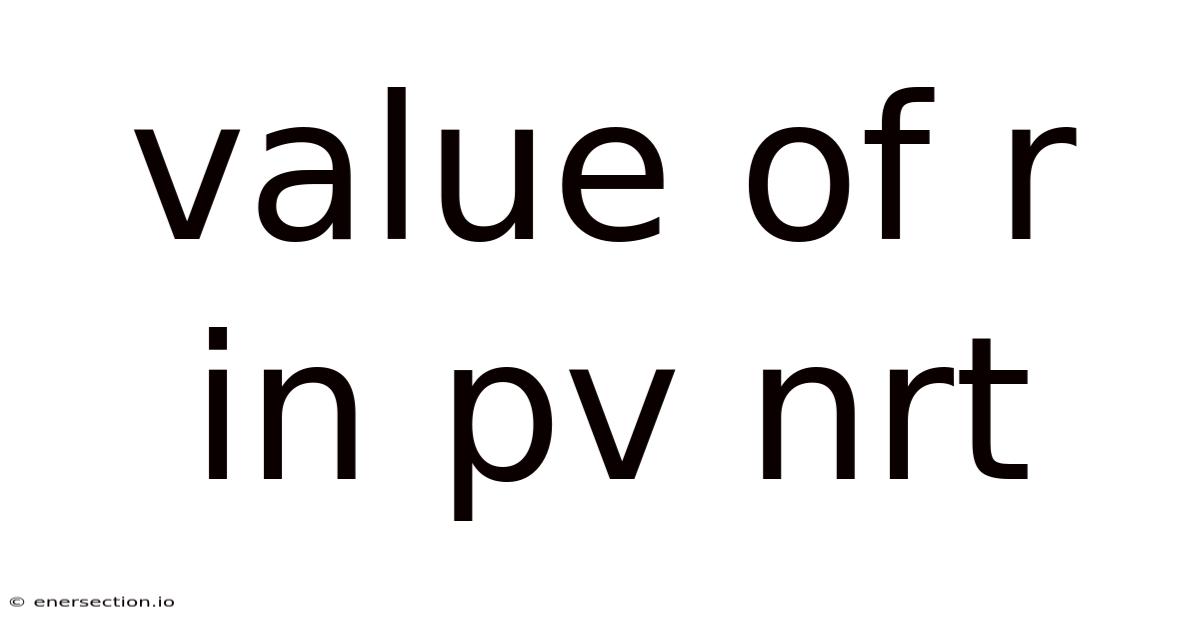The ideal gaslaw, expressed as PV = nRT, is a cornerstone equation that links pressure (P), volume (V), amount of substance (n), temperature (T), and the gas constant (R). Understanding the value of r in PV nRT is essential for anyone studying thermodynamics, chemistry, engineering, or related fields, because R serves as the bridge that converts between macroscopic gas properties and microscopic behavior. This article explores the nature of R, its typical values, how to select the appropriate unit, practical calculation steps, and common pitfalls, providing a practical guide that can be used for study, reference, or SEO‑optimized content Still holds up..
Not obvious, but once you see it — you'll see it everywhere It's one of those things that adds up..
What is the Gas Constant R?
The gas constant R is a universal constant that appears in the ideal gas equation PV = nRT. It represents the proportionality factor that relates the energy scale in an ideal gas to its temperature. Consider this: in essence, R quantifies how much work or heat is needed to increase the temperature of a given amount of gas by one degree. Although R is a constant, its numerical value changes depending on the units used for pressure, volume, and temperature.
Key points about R
- R is universal: it applies to all ideal gases, regardless of chemical identity.
- R is derived: it originates from the combination of Boyle’s law, Charles’s law, Avogadro’s law, and Gay‑Lussac’s law.
- R is dimensionful: its units reflect the combination of energy per mole per kelvin.
Common Units of R
Because R can be expressed in many unit systems, it is crucial to match the units of P, V, n, and T consistently. Below are the most frequently used values:
| Units for P | Units for V | Units for n | Units for T | Value of R |
|---|---|---|---|---|
| Atmospheres (atm) | Liters (L) | Moles (mol) | Kelvin (K) | 0.314462 J·mol⁻¹·K⁻¹ |
| Millimeters of mercury (mm Hg) | Liters (L) | Moles (mol) | Kelvin (K) | 62.In real terms, 082057 L·atm·mol⁻¹·K⁻¹ |
| Pascals (Pa) | Cubic meters (m³) | Moles (mol) | Kelvin (K) | 8. 3637 L·mm Hg·mol⁻¹·K⁻¹ |
| Bars | Cubic meters (m³) | Moles (mol) | Kelvin (K) | **0. |
Why unit consistency matters: If you plug pressure in atmospheres but volume in cubic meters, the calculated R will be incorrect, leading to erroneous results. Always convert all quantities to a compatible set before using the equation.
How to Determine Which R Value to Use
- Identify the pressure unit you will use (e.g., atm, Pa, mm Hg).
- Identify the volume unit (e.g., L, m³).
- Select the temperature scale (Kelvin is mandatory; Celsius must be converted to Kelvin by adding 273.15).
- Choose the corresponding R value from the table above.
Example: If you are working with pressure in kilopascals (kPa) and volume in cubic meters, you would use R = 8.314 J·mol⁻¹·K⁻¹, because 1 J = 1 Pa·m³, and kPa·m³ = 1 kJ, preserving unit consistency Simple as that..
Practical Steps for Calculating R in PV = nRT
Below is a step‑by‑step workflow that can be applied to laboratory experiments, industrial calculations, or exam problems Not complicated — just consistent..
- Write the ideal gas equation: PV = nRT.
- Gather known quantities:
- Pressure (P) – ensure it is in the chosen unit. - Volume (V) – ensure it matches the unit selected for R.
- Amount of substance (n) – usually measured in moles.
- Temperature (T) – convert to Kelvin if necessary. 3. Select the appropriate R value based on the unit set from step 2.
- Rearrange the equation to solve for the unknown variable.
- To find P: P = nRT / V
- To find V: V = nRT / P
- To find n: n = PV / (RT)
- To find T: T = PV / (nR) 5. Insert the numbers and perform the calculation.
- Check units: The resulting unit should match the expected unit for the variable you solved for (e.g., atm for pressure, L for volume, mol for amount).
- Validate the result by considering physical plausibility (e.g., a temperature of 300 K is reasonable for room‑temperature gases).
Illustrative example:
Suppose you have 0.5 mol of an ideal gas at 2 atm pressure and a volume of 10 L. To find the temperature:
- Use R = 0.082057 L·atm·mol⁻¹·K⁻¹.
- Rearrange: T = PV / (nR) = (2 atm × 10 L) / (0.5 mol × 0.082057 L·atm·mol⁻¹·K⁻¹).
- Compute: T ≈ (20) / (0.0410285) ≈ 487 K.
The result indicates a temperature of about 487 K (≈214 °C), which aligns with the given conditions.
Scientific Explanation Behind R
The gas constant R emerges from the combination of several fundamental gas laws:
- Boyle’s Law: At constant temperature, P is inversely proportional to V (PV