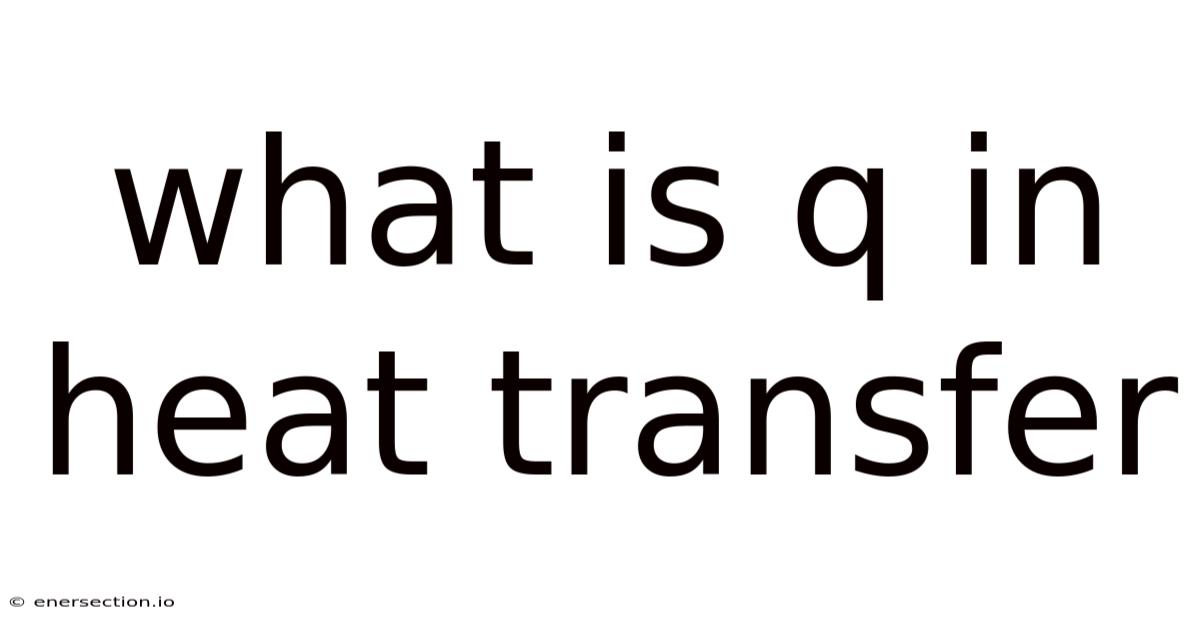What Is “q” in Heat Transfer? Understanding the Symbol, Its Meaning, and Its Applications
In the realm of thermal sciences, the symbol q appears in almost every textbook, research paper, and engineering calculation related to heat transfer. It represents the rate of heat transfer, a fundamental quantity that describes how much thermal energy moves from one place to another per unit time. Grasping what q stands for, how it is measured, and how it fits into the governing equations is essential for students, researchers, and practicing engineers who work with heating, cooling, and energy‑conversion systems The details matter here. Surprisingly effective..
Introduction: Why the Symbol “q” Matters
Heat transfer governs everything from the temperature of a cup of coffee cooling on a desk to the efficiency of a power plant’s turbine. Practically speaking, the heat transfer rate (q) quantifies the speed at which thermal energy is exchanged, allowing designers to size heat exchangers, predict temperature profiles, and evaluate the performance of insulation. Because q directly links energy, time, and temperature, it serves as the bridge between theoretical analysis and real‑world applications.
Defining q: The Basic Concept
- Physical Meaning: q is the amount of heat energy transferred per unit time. Its SI unit is the watt (W), which is equivalent to joules per second (J·s⁻¹).
- Mathematical Form:
[ q = \frac{dQ}{dt} ]
where dQ is an infinitesimal amount of heat and dt is an infinitesimal time interval.
When the heat transfer is steady (i.e., does not change with time), the differential notation collapses to a simple ratio:
[ q = \frac{Q}{t} ]
The Three Modes of Heat Transfer and Their q‑Expressions
1. Conduction
Conduction occurs when heat moves through a solid material due to a temperature gradient. Fourier’s law provides the relationship:
[ \boxed{q = -k A \frac{dT}{dx}} ]
- k – thermal conductivity of the material (W·m⁻¹·K⁻¹)
- A – cross‑sectional area normal to the heat flow (m²)
- dT/dx – temperature gradient in the direction of heat flow (K·m⁻¹)
The negative sign indicates that heat flows from higher to lower temperature But it adds up..
2. Convection
Convection describes heat transfer between a solid surface and a moving fluid (liquid or gas). Newton’s law of cooling defines the rate:
[ \boxed{q = h A (T_s - T_\infty)} ]
- h – convective heat transfer coefficient (W·m⁻²·K⁻¹)
- T_s – surface temperature of the solid (K)
- T_\infty – bulk fluid temperature far from the surface (K)
The coefficient h encapsulates complex phenomena such as fluid velocity, viscosity, and turbulence.
3. Radiation
Thermal radiation is energy emitted as electromagnetic waves. The Stefan‑Boltzmann law gives:
[ \boxed{q = \varepsilon \sigma A (T_s^4 - T_{\text{sur}}^4)} ]
- \varepsilon – emissivity of the surface (dimensionless, 0–1)
- \sigma – Stefan‑Boltzmann constant (5.670 × 10⁻⁸ W·m⁻²·K⁻⁴)
- T_{\text{sur}} – temperature of the surrounding environment (K)
Radiative heat transfer becomes dominant at high temperatures because of the fourth‑power temperature dependence.
Units, Sign Conventions, and Directionality
| Quantity | Symbol | Unit | Typical Sign Convention |
|---|---|---|---|
| Heat transfer rate | q | Watt (W) | Positive when heat leaves the control volume (or enters a system, depending on context) |
| Heat flux (per unit area) | q″ | W·m⁻² | Positive in the direction of heat flow; often written as q″ = q/A |
| Thermal conductivity | k | W·m⁻¹·K⁻¹ | Always positive for normal materials |
| Convective coefficient | h | W·m⁻²·K⁻¹ | Positive; larger values indicate more efficient convection |
In many engineering texts, q is taken as positive when heat enters the system (e.g., a heater supplying energy) and negative when heat leaves (e.Worth adding: g. , cooling). Consistency within a problem is crucial; otherwise, sign errors can lead to unrealistic temperature predictions.
Some disagree here. Fair enough The details matter here..
Deriving q from Energy Balances
A typical method to find q involves setting up an energy balance on a control volume. Consider a one‑dimensional slab of thickness L, steady‑state conduction, and constant thermal conductivity k. The energy balance states that the heat entering one face equals the heat leaving the opposite face:
[ q_{\text{in}} = q_{\text{out}} = q ]
Integrating Fourier’s law across the slab yields:
[ q = \frac{k A (T_1 - T_2)}{L} ]
where T₁ and T₂ are the temperatures at the two faces. This simple expression demonstrates how q scales with temperature difference, material conductivity, and geometry And that's really what it comes down to..
In more complex geometries (cylinders, spheres), the same principle applies but the area A varies with position, leading to modified forms such as:
[ q = \frac{2 \pi k L (T_{\text{inner}} - T_{\text{outer}})}{\ln(r_{\text{outer}}/r_{\text{inner}})} \quad \text{(cylindrical wall)} ]
Practical Applications of q
Heat Exchangers
Designers calculate q to size the tubes, fins, and flow rates that achieve a desired thermal duty. The overall heat transfer coefficient U combines conduction, convection, and sometimes radiation:
[ q = U A_{\text{LM}} \Delta T_{\text{LM}} ]
where A_{\text{LM}} is the logarithmic mean area and ΔT_{\text{LM}} is the logarithmic mean temperature difference between the hot and cold streams Less friction, more output..
Building Insulation
In building physics, q through walls and roofs determines heating and cooling loads. By selecting insulation with low k, architects reduce q, thereby lowering energy consumption.
Electronics Cooling
Microprocessors generate significant heat. The cooling solution must remove a q that can reach hundreds of watts per square centimeter. Engineers select high‑h fans or liquid‑cooling loops to meet the required q Small thing, real impact..
Aerospace and High‑Temperature Processes
Radiative heat transfer dominates during spacecraft re‑entry or furnace operation. Accurate q predictions using the Stefan‑Boltzmann law are essential for material selection and safety Simple, but easy to overlook..
Frequently Asked Questions (FAQ)
Q1: Is q the same as heat energy Q?
No. Q denotes the total amount of heat transferred (Joules), while q is the rate at which that heat moves (Watts). Think of Q as the distance traveled and q as the speed.
Q2: Can q be negative?
Yes. The sign of q depends on the chosen reference direction. If heat flows opposite to the defined positive direction, q becomes negative, indicating a reversal of energy flow Worth knowing..
Q3: How does the concept of heat flux q″ differ from q?
q″ is the heat transfer rate per unit area (W·m⁻²). It provides a local measure of heat flow, useful for non‑uniform surfaces. The relationship is q = q″ · A when the area is uniform Easy to understand, harder to ignore..
Q4: Why does radiation depend on the fourth power of temperature?
The Stefan‑Boltzmann law arises from quantum mechanics and Planck’s radiation theory. It shows that as temperature rises, the spectrum shifts to higher frequencies, dramatically increasing emitted energy.
Q5: What factors influence the convective coefficient h?
Surface roughness, fluid velocity, fluid properties (viscosity, density, specific heat), and flow regime (laminar vs. turbulent) all affect h. Empirical correlations (e.g., Nusselt number relations) are commonly used to estimate it But it adds up..
Solving a Sample Problem: Heat Transfer Through a Wall
Given: A wall 0.2 m thick, area 10 m², thermal conductivity k = 0.04 W·m⁻¹·K⁻¹. Inside temperature T_i = 22 °C, outside temperature T_o = -5 °C.
Find: The steady‑state heat transfer rate q.
Solution
-
Convert temperatures to Kelvin (optional, but temperature difference is the same):
ΔT = 22 °C – (‑5 °C) = 27 K Small thing, real impact. No workaround needed.. -
Apply the one‑dimensional conduction formula:
[ q = \frac{k A \Delta T}{L} ] -
Insert the values:
[ q = \frac{0.04 \times 10 \times 27}{0.2} = \frac{10.8}{0.2} = 54 \text{ W} ]
Thus, the wall conducts 54 W of heat from the interior to the exterior. If the goal were to reduce this heat loss, increasing L (adding insulation) or decreasing k (using a lower‑conductivity material) would lower q Not complicated — just consistent..
Common Mistakes to Avoid
- Mixing up q and q″ – Always verify whether you need total heat rate or heat flux per area.
- Neglecting Contact Resistances – In multilayer assemblies, thermal contact resistance can dominate, effectively adding an extra term to the overall q calculation.
- Assuming Constant Properties – Thermal conductivity k often varies with temperature; for large ΔT, integrate k(T) across the material.
- Ignoring Radiation in High‑Temp Scenarios – At temperatures above ~500 K, radiative heat can exceed conduction and convection, and must be included in the energy balance.
Conclusion: The Central Role of q in Thermal Engineering
The symbol q encapsulates the essence of heat transfer: how fast thermal energy moves. Practically speaking, whether expressed through Fourier’s law for conduction, Newton’s law for convection, or the Stefan‑Boltzmann law for radiation, q provides a quantitative link between material properties, geometry, temperature differences, and fluid dynamics. Mastery of the concept enables engineers to design efficient heating and cooling systems, improve energy conservation in buildings, safeguard electronic components, and predict the behavior of high‑temperature processes. By understanding the underlying physics, applying correct sign conventions, and carefully handling units, practitioners can avoid common pitfalls and produce reliable, real‑world solutions that stand up to the rigorous demands of modern thermal engineering The details matter here..