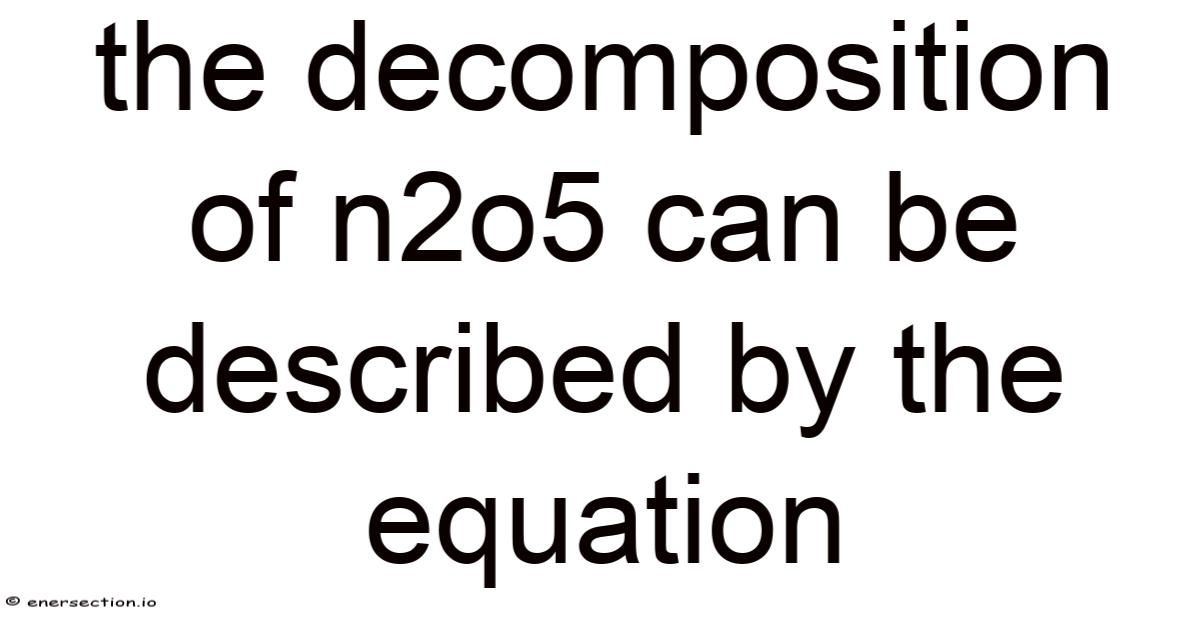Thedecomposition of N2O5 is a fundamental chemical reaction that serves as a cornerstone in understanding reaction kinetics and gas-phase processes. This reaction involves the breakdown of dinitrogen pentoxide (N2O5), a compound composed of nitrogen and oxygen, into simpler molecules. Still, the decomposition of N2O5 is not only a classic example in chemistry education but also a critical process in atmospheric chemistry and industrial applications. But the reaction is typically represented by the balanced equation: 2N2O5 → 4NO2 + O2. Worth adding: this transformation highlights the dynamic nature of chemical systems and the interplay between reactants and products under specific conditions. Understanding the decomposition of N2O5 provides insights into how complex molecules can break down into simpler components, a principle that applies to numerous real-world scenarios. The study of this reaction also underscores the importance of reaction mechanisms, rate laws, and the role of catalysts in influencing chemical behavior. By examining the decomposition of N2O5, chemists can better grasp the principles governing chemical stability and reactivity, which are essential for advancing both theoretical and applied chemistry.
Not obvious, but once you see it — you'll see it everywhere It's one of those things that adds up..
The decomposition of N2O5 follows a well-defined reaction pathway, which can be analyzed through both experimental observations and theoretical models. In practice, the decomposition process is typically initiated by thermal energy, as N2O5 is relatively stable under standard conditions but becomes reactive when subjected to elevated temperatures. In real terms, the decomposition of N2O5 is particularly notable for its first-order kinetics, meaning the rate of reaction depends solely on the concentration of N2O5. Here's the thing — this stoichiometric relationship is crucial for predicting the quantities of products formed during the reaction. Additionally, the reaction is often studied in a closed system to monitor changes in concentration over time, allowing researchers to determine the reaction order and rate constant. On the flip side, when N2O5 is heated or exposed to certain conditions, it undergoes a series of steps that lead to the formation of nitrogen dioxide (NO2) and oxygen gas (O2). Consider this: the reaction rate is influenced by factors such as temperature, pressure, and the presence of catalysts. The balanced equation, 2N2O5 → 4NO2 + O2, indicates that two molecules of N2O5 decompose to produce four molecules of NO2 and one molecule of O2. Take this case: higher temperatures generally increase the rate of decomposition, as they provide the necessary activation energy for the reaction to proceed. This characteristic makes it a valuable model for studying reaction kinetics and validating theoretical predictions Practical, not theoretical..
The mechanism of N2O5 decomposition involves several intermediate steps that are not immediately obvious from the balanced equation. This oxygen can influence the surrounding environment, particularly in atmospheric contexts where N2O5 is a significant source of NO2. These intermediate species play a key role in the overall reaction, even though they are not present in the final products. In real terms, the decomposition of N2O5 also involves the release of oxygen gas, which is a byproduct of the reaction. The nitronium ion then reacts with another NO2 molecule to form nitrogen trioxide (N2O3), which further decomposes into NO and NO2. One widely accepted mechanism begins with the dissociation of N2O5 into nitrogen dioxide (NO2) and a nitronium ion (NO3+). The formation and subsequent breakdown of these intermediates illustrate the complexity of chemical reactions and the importance of understanding reaction mechanisms. As an example, the nitronium ion (NO3+) is a highly reactive species that can participate in various chemical processes, including the formation of ozone or other nitrogen oxides. This initial step is critical because it sets the stage for subsequent reactions. The interplay between these molecules highlights the interconnectedness of chemical processes and their broader implications Which is the point..
The decomposition of N2O5 is not only a theoretical exercise but also has practical applications in various fields. In atmospheric chemistry, N2O5 is a key precursor to nitrogen dioxide (NO2), which plays a role in the formation of photochemical smog. The decomposition of N2O5 in the atmosphere contributes to the production of NO2, a pollutant that can have adverse effects on air quality and human health. Understanding the kinetics and mechanisms of this reaction is essential for modeling atmospheric processes and developing strategies to mitigate pollution.
The practical utility of N2O5 decomposition extends into industrial chemistry, where it serves as a critical intermediate in the synthesis of nitrogen-based compounds. To give you an idea, controlled decomposition processes are employed in the production of nitric acid, a cornerstone of fertilizer manufacturing. By manipulating reaction conditions such as pressure and catalyst presence, industries can optimize yield and minimize waste, showcasing the reaction’s adaptability to large-scale applications. On top of that, additionally, the study of N2O5 decomposition has informed advancements in catalysis, as researchers seek to design materials that can either accelerate or inhibit such reactions depending on the desired outcome. Catalysts like metal oxides or zeolites have shown promise in modulating reaction pathways, offering insights that could revolutionize energy storage or emission control technologies But it adds up..
Beyond industrial uses, the decomposition of N2O5 plays a role in environmental monitoring and remediation. Still, scientists take advantage of its predictable kinetics to develop sensors that detect trace amounts of nitrogen oxides in air and water samples, aiding in pollution tracking and regulatory compliance. Beyond that, the reaction’s sensitivity to external factors—such as humidity or the presence of other gases—makes it a valuable tool for probing atmospheric dynamics in climate models. Recent studies have also explored its potential in green chemistry, where understanding decomposition pathways could lead to more sustainable methods for breaking down hazardous nitrogen compounds It's one of those things that adds up..
Looking ahead, the decomposition of N2O5 continues to inspire interdisciplinary research. Think about it: emerging fields like nanotechnology and computational chemistry are shedding new light on reaction mechanisms at the molecular level, enabling precise predictions about how structural modifications might alter reactivity. These developments not only deepen our understanding of fundamental chemistry but also pave the way for innovations in energy conversion, environmental protection, and materials science Not complicated — just consistent..
At the end of the day, the decomposition of N2O5 stands as a compelling example of how a single chemical process can bridge theoretical inquiry and real-world application. And its study has enriched our grasp of reaction kinetics, illuminated atmospheric processes, and driven technological progress across multiple sectors. As research advances, the lessons learned from this reaction will undoubtedly contribute to solving complex challenges in sustainability and environmental stewardship, underscoring the enduring value of foundational scientific exploration.
The complex dance of molecular transformations thus holds profound significance across countless domains. Future research promises even greater efficiencies and novel pathways, pushing boundaries further. Such endeavors underscore the constant interplay between fundamental science and practical utility.
In closing, understanding decomposition like N2O5 remains critical, offering insights vital for responsible technological development and sustainable progress That's the part that actually makes a difference..
So, continued study remains essential, ensuring knowledge is applied wisely. Its legacy endures.
The evolving field continues to unveil new possibilities, each discovery weaving into the broader tapestry of scientific progress. As methodologies advance, their impact becomes increasingly nuanced, demanding careful consideration. Collaborative efforts now bridge theoretical and applied realms, fostering innovations that address global challenges. Such dynamism underscores the importance of staying attuned to shifts in research focus That's the part that actually makes a difference. Simple as that..
In closing, the interplay between curiosity and application remains central, reminding us that understanding often precedes transformation. Such cycles highlight the enduring relevance of foundational studies, shaping futures both tangible and abstract. Their legacy persists, a testament to the power of sustained inquiry Worth knowing..